Figures & data
Table 1. Characteristic of patients at baseline (FAS).
Table 2. The summary of primary endpoint.
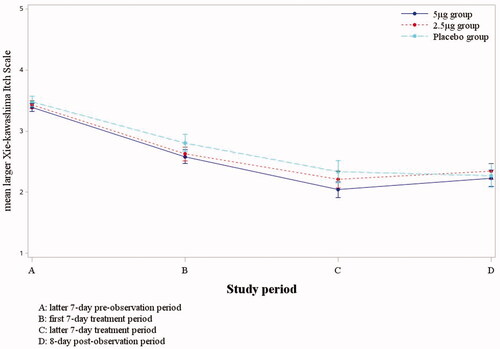
Table 3. The summary change of mean larger Xie-kawashima itch scale between latter seven-day pre-observation and latter seven-day treatment period.
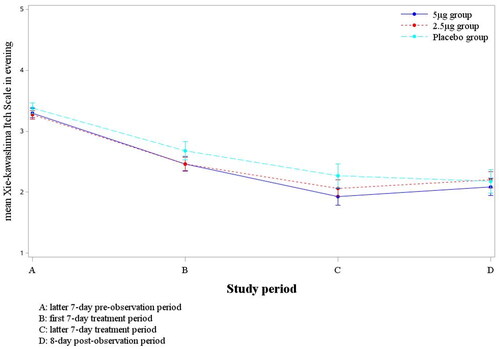
Table 4. The summary change of mean Xie-kawashima itch scale in evening between latter seven-day pre-observation and latter seven-day treatment period.
Table 5. The summary of improvement of VAS value and effect.
Table 6. The summary of adverse events.