Figures & data
Figure 1. Schematic of the experimental apparatus. A continuous supply of artificial urine medium (AUM) is provided to an artificial bladder using a peristaltic pump (flow rate = 0.5 mm min–1). The top portion of the catheter is inserted into an artificial bladder while the bottom portion is connected to tubing that drains into a waste container. The entire apparatus is enclosed in a modified incubator at 37°C. Arrows symbolize the direction of medium flow.
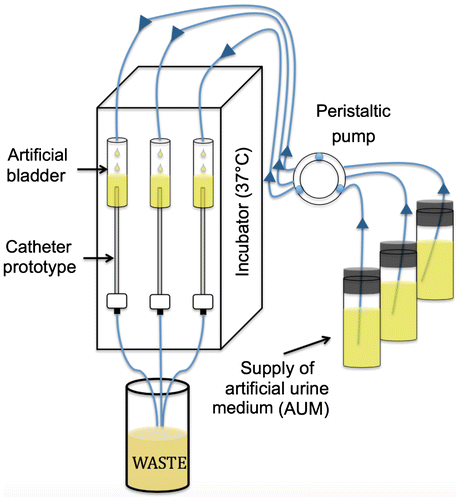
Figure 2. Representative optical images of C. albicans biofilms grown in catheter prototypes compared to a negative control (A). Cross sectional optical images of biofilm growth within the main lumen at different regions along the length of the catheter prototype (B). Scale bars = 1 cm (upper) and 1 mm (lower).

Figure 3. SEM images of C. albicans biofilms formed inside catheters. (A) 63× magnification with 30 kV and scale bar = 1 mm. (B) 1,000× magnification, 5 kV, scale bar = 50 μm. (C) 4,000× magnification, 5 kV, scale bar = 10 μm. (D) 10,000× magnification, 5 kV, scale bar = 5 μm.
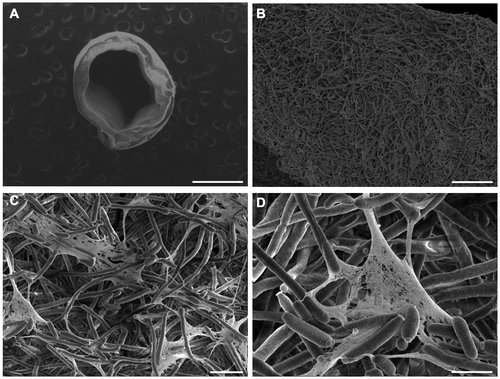
Figure 4. Tensile testing of intact C. albicans biofilms formed in catheter prototypes. (A) Biofilms were manually extracted from catheter prototypes and mounted in a microstrain analyzer for tensile testing. (B) Representative stress–strain plot resulting from tensile testing of intact biofilms.
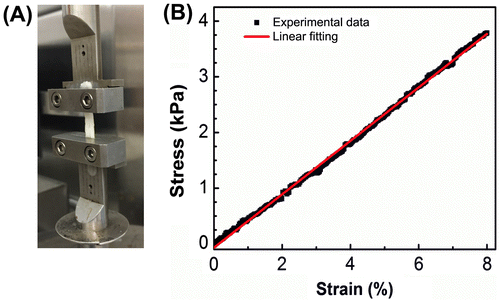
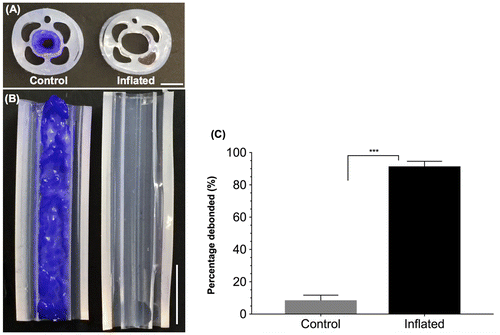
Figure 5. Catheter prototypes debond C. albicans biofilms. Representative cross sectional (A) and longitudinal (B) optical images of control (not inflated, left) and debonded (inflated, right) biofilms in the main lumen of the catheter prototypes. Biofilms were stained with crystal violet to enhance visualization. Scale bars = 2 mm (A) and 5 mm (B). (C) Percentage biofilm mass removed from the control and inflated catheters (N = 4). ‘***’ indicates p < 0.001.