Figures & data
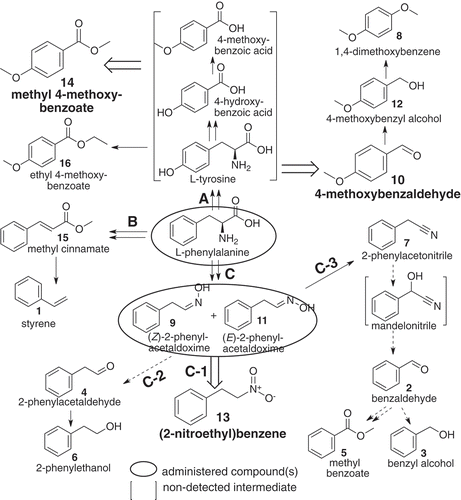
Figure 1. Photograph of panicle incubations {left: new flower (NF), right: old flower (OF)} with d8-L-phenylalanine administered as aqueous solution in vials. Arrow indicates a mono trap RCC18 hanged with steal wire. After setting the trap to both shoots, both flowers were covered by polyethylene bags to collect flower scent volatiles.

Figure 2. Typical GC profile of flower scents volatiles by mono trap RCC18, emanated from old flower panicle (OF) treated with d8-L-phenylalanine during 24–48 h (p2), and each peak analyses by SIC to assess d-labeled isomer content. Solid line: natural component, dotted line: d-labeled isomer.
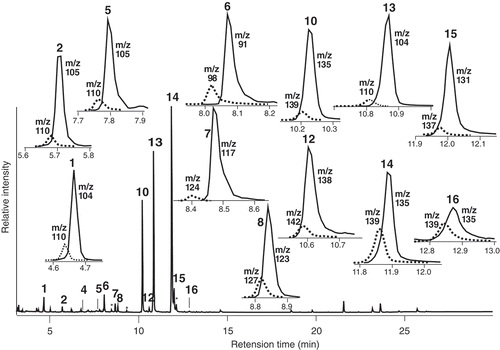
Figure 3. Typical GC profile of hexane extracts after incubation of a flower petal overnight with d5-2-phenylacetaldoximes (d5-9 & −11), and each peak analyses by SIC to assess d-labeled isomer content. Solid line: natural component, Dotted line: d-labeled isomer.
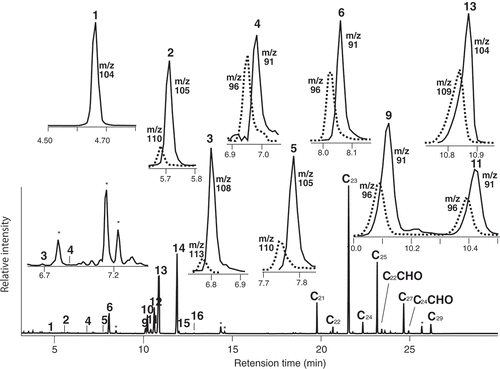
Table 1. Gas chromatographic and mass spectral data of flower scent compounds from Japanese loquat Eriobotrya japonica after incubation by d8-L-phenylalamnine and by a mixture of d5-Z- and -E-2-phenylacetaldoxime.
Table 2. d-Labeled content (%) of flower scents from Loquat after incubation with d8-L-phenylalanine.
Table 3. Major components of hexane extracts after incubation of flower petal by d5-9 & −11.
Figure 4. A proposed pathway of (2-nitroethyl)benzene and other related benzenoids detected in loquat flower scents, derived from L-phenylalanine by way of Z- and E-2-phenylacetaldoxime. Compounds in blanket were not detected in the present methods. Broad open arrows indicate reactions to major components. Solid line: reaction pathway(s) reported or conceivable, doted line: suggestive pathway, including non-enzymic step(s) partly.
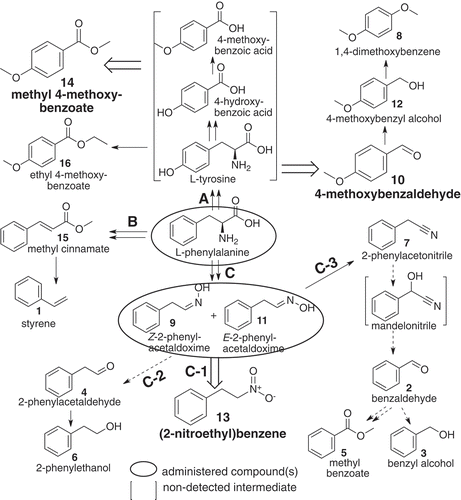
Table 4. Distribution of (2-nitroethyl)benzene (Nitro), 2-phenylacetaldoximes (Oxime) and 2-phenylacetonitrile (Nitrile) in the plant kingdom as components of flower scents (El-Sayed AM 2017).
