Figures & data
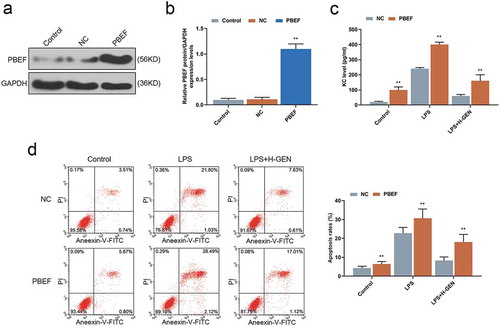
Figure 1. Effects of genistein on viability of MLE-12 cells.
(a) Low concentration of genistein (1–10 µM) did not affect cell viability, while 25 µM genistein inhibited cell viability by spectrophotometric method 3-[4,5-dimethylthiazol-2-yl]-2,5-diphenyltetrazolium bromide (MTT) experiment. **p < 0.001 vs. genistein (0 µM). (b) High concentration of genistein did not affect cell viability, lipopolysaccharide (LPS) inhibited cell viability, PBEF inhibitor FK866 induces increased cell viability and genistein enhanced LPS-induced low cell viability in a concentration-dependent manner by MTT experiment. **p < 0.001 vs. Control; ##p < 0.001 vs. LPS; ^^p < 0.001 vs. LPS + L-GENISTEIN; ΦΦP<0.001 vs. LPS + H-GENISTEIN.
![Figure 1. Effects of genistein on viability of MLE-12 cells.(a) Low concentration of genistein (1–10 µM) did not affect cell viability, while 25 µM genistein inhibited cell viability by spectrophotometric method 3-[4,5-dimethylthiazol-2-yl]-2,5-diphenyltetrazolium bromide (MTT) experiment. **p < 0.001 vs. genistein (0 µM). (b) High concentration of genistein did not affect cell viability, lipopolysaccharide (LPS) inhibited cell viability, PBEF inhibitor FK866 induces increased cell viability and genistein enhanced LPS-induced low cell viability in a concentration-dependent manner by MTT experiment. **p < 0.001 vs. Control; ##p < 0.001 vs. LPS; ^^p < 0.001 vs. LPS + L-GENISTEIN; ΦΦP<0.001 vs. LPS + H-GENISTEIN.](/cms/asset/5ca6cee9-b2b4-495b-bf02-b8c9749d0202/tbbb_a_1697197_f0001_oc.jpg)
Figure 2. Effects of genistein on inflammatory factors and apoptosis of MLE-12 cells.
(a–d) High concentration of genistein treatment alone had no significant effect on the levels of inflammatory factors. LPS increased the contents of inflammatory factors, FK866 induced decreased contents of inflammatory factors and genistein decreased LPS-induced high levels of inflammatory factors in a concentration-dependent manner by enzyme-linked immunosorbent assay (ELISA). (e) High concentration of genistein alone had no significant effect on apoptosis of MLE-12 cells. LPS increased the apoptotic rate, FK866 induced decreased cell apoptotic rate and genistein decreased the apoptotic rate of MLE-12 cells in a concentration-dependent manner by flow cytometry test. **p < 0.001 vs. Control; ##p < 0.001 vs. LPS; ^^p < 0.001 vs. LPS + L-GENISTEIN; ΦΦP<0.001 vs. LPS + H-GENISTEIN.
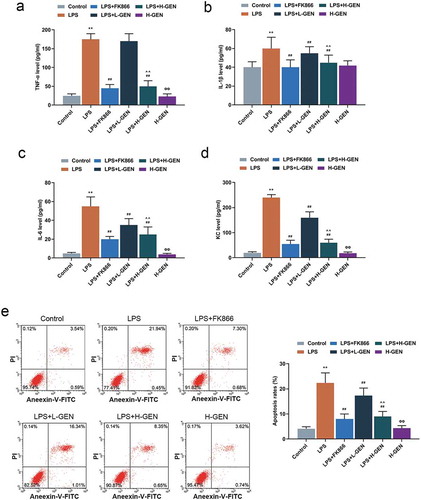
Figure 3. Effects of genistein on PBEF and NF-κB pathway protein.
(a) High concentration of genistein treatment alone had no significant effect on PBEF expression. LPS up-regulated the expression of PBEF, while high concentration of genistein and FK866 decreased PBEF expression by western blot (WB) analysis. (b–c) High concentration of genistein treatment alone had no significant effect on p65 expression. LPS up-regulated nuclear p65 and down-regulated cytoplasmic p65, H-GENISTEIN and FK866 down-regulated nuclear p65 and up-regulated cytoplasmic p65 western blotting analysis. **p < 0.001 vs. Control; ##p < 0.001 vs. LPS; ΦP<0.05, ΦΦP<0.001 vs. LPS + H-GENISTEIN.
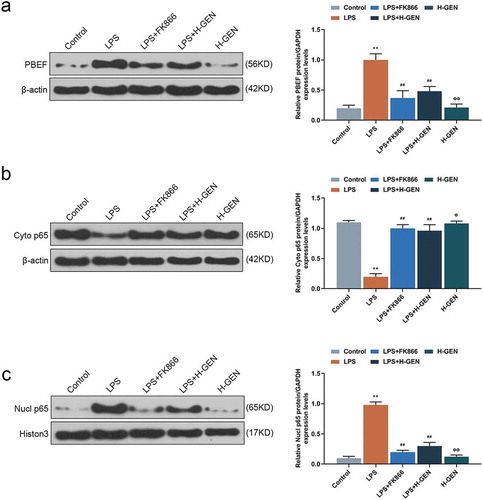
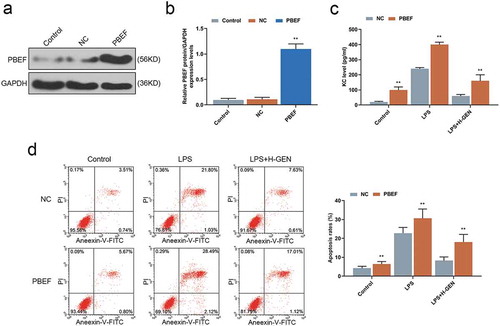
Figure 4. Effects of genistein on keratinocyte-derived chemokine (KC) and MLE-12 cells apoptosis via regulating PBEF.
(a–b) WB test showed that PBEF transfection was successful. (c) Overexpression of PBEF attenuated the inhibitory effect of genistein on KC release. (d) Overexpression of PBEF attenuated the inhibitory effect of GENISTEIN on MLE-12 cell apoptosis. **p < 0.001 vs. NC.