Figures & data
Table I. Baseline clinical characteristics of patients with chronic hepatitis C (n = 28).
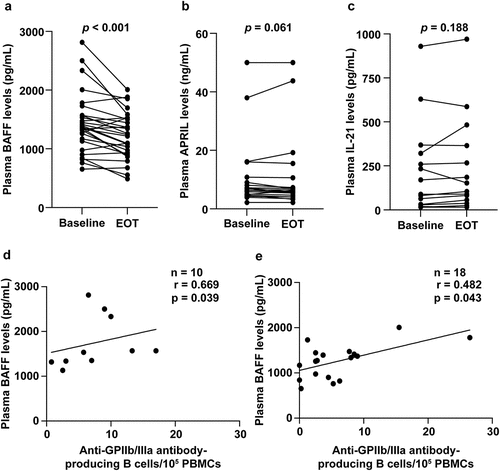
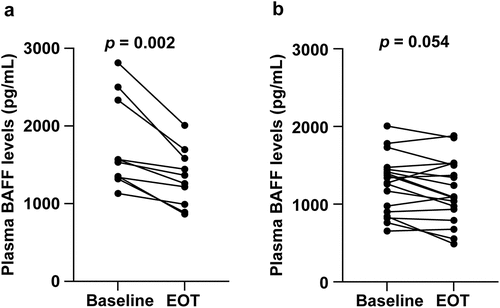
Table II. Changes in laboratory findings and thrombopoietin levels at baseline and end of treatment (EOT) with direct-acting antiviral agents (DAA).
Figure 1. Frequency of anti-GPIIb/IIIa antibody-producing cells in patients with chronic hepatitis C with thrombocytopenia (CHC TP+), patients with CHC without thrombocytopenia (CHC TP−), and healthy controls. Horizontal bars represent the median values; p-values <0.05 were considered significant.
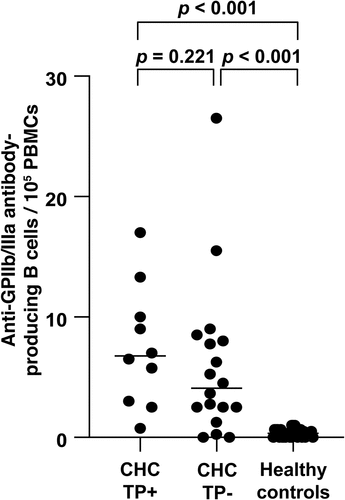
Figure 2. Changes in the (a) platelet count and (b) number of anti-GPIIb/IIIa antibody-producing cells before and after DAA treatment. (c) Enzyme-linked immunospot data for anti-GPIIb/IIIa antibody-producing cells at baseline and after DAA therapy are shown.
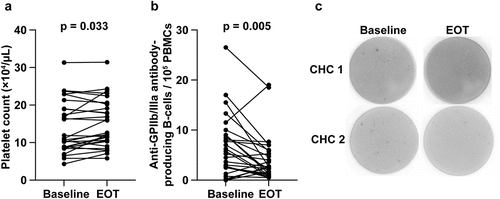
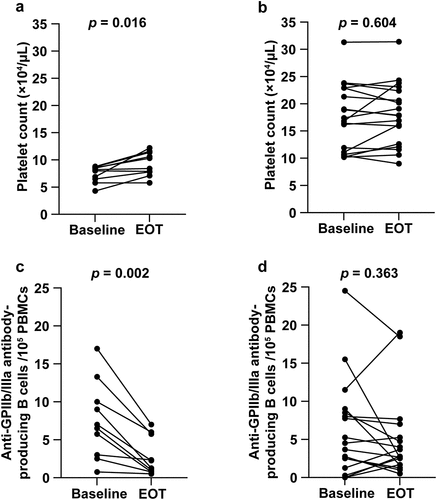
Figure 3. Changes in the (a, b) platelet count and (c, d) number of anti-GPIIb/IIIa antibody-producing cells after DAA treatment. Groups (a, c) with and (b, d) without thrombocytopenia at baseline.