Figures & data
Figure 1. Subject disposition. Three analysis populations were defined for the study: full-analysis-set (FAS, including all subjects injected at least once), per protocol set (PPS, including all subjects who completed all planned treatments without major protocol deviations), and safety set (SS, including all subjects who received injections and underwent at least one post-treatment safety assessment).
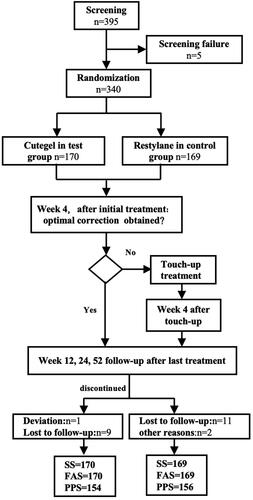
Table 1. Baseline demographics and characteristics (FAS).
Table 2. Response rates and differences in response rates based on the WSRS (at least one-point improvement) at week 24 by a blinded evaluator.
Figure 2. Comparison of response rates based on the WSRS, as assessed by a blinded evaluator during follow-up.
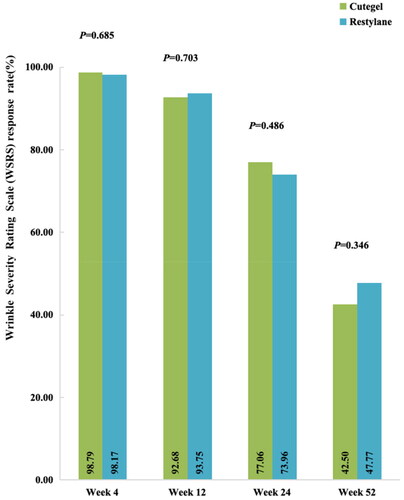
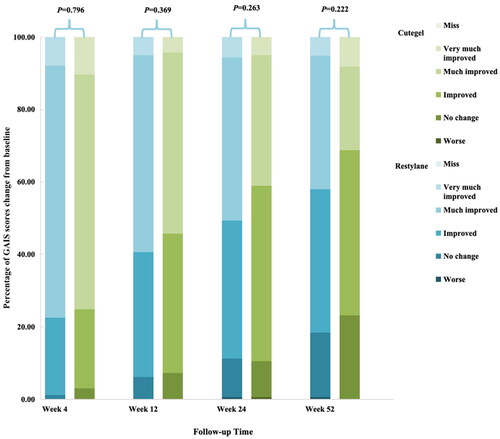
Figure 3. Comparison of response rates based on the GAIS as assessed by a blinded evaluator during follow-up.
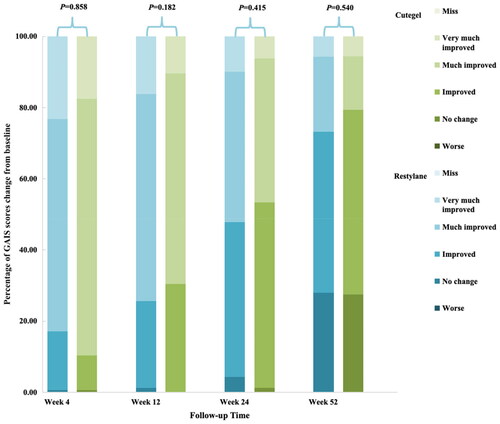
Table 3. Incidence of treatment site responses.
Data availability statement
Data are available on reasonable requests from the corresponding author.