Figures & data
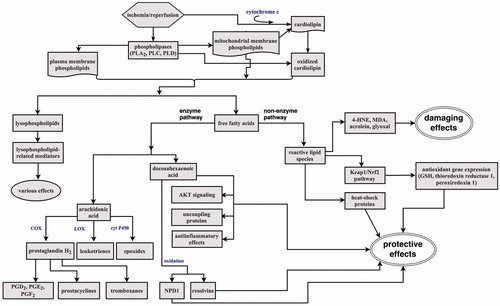
Figure 1. Cascade of molecular events initialized by ischemia/reperfusion. The etiology of ischemic-reperfusion injury involves bioenergetic failure, the loss of intracellular ion homeostasis, membrane integrity, glutamate excitotoxicity, protein synthesis inhibition, progressive proteolysis, inflammation, oxidative stress, and reduced survival signal transduction (more details in the text). NOS: nitric oxide synthase; ER: endoplasmic reticulum; eIF2: eukaryotic initiation factor 2; ROS: reactive oxygen species; Bax: Bcl-2-associated X protein; Puma: p53 upregulated modulator of apoptosis; Noxa: PMA-induced protein.
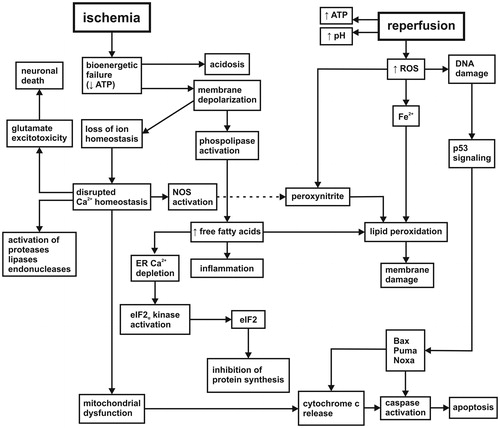
Figure 2. Preferential utilization of glucose in neurons. Neurons preferentially utilize glucose in the pentose phosphate pathway to produce reduced glutathione. Under energy crisis glucose is redirected into glycolysis to produce ATP resulting in weakened antioxidant defense (see the text for more details). TCA: tricarboxylic acid cycle; IF1: F1F0-ATPase inhibitory factor 1; DHAP: dihydroxyacetone phosphate.
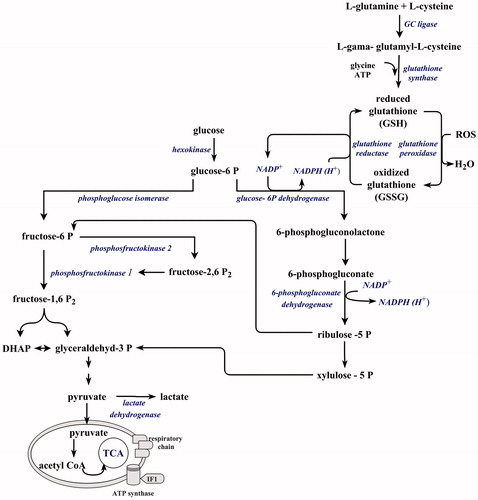
Figure 3. Ischemia/reperfusion-induced breakdown of membrane phospholipids. Activation of phospholipases triggers phospholipid hydrolysis releasing lysophospholipids and free fatty acids. Free fatty acids undergo oxidative metabolism by enzyme and non-enzyme pathways. The enzyme-catalyzed pathways produce oxygenated derivatives such as prostaglandines, prostacyclines, tromboxanes, leukotriens, or epoxides from arachidonic acid. They are responsible for postischemic inflammation. DHA oxoderivatives can elicit antiinflammatory and protective effects. Oxoderivatives of non-enzyme pathways produce reactive lipid species that can be involved in both damaging and protective cell responses (more details in the text). PGD2, PGE2, PGF2: prostaglandins; NPD1: neuroprotectin 1; 4-HNE: 4-hydroxy-2-nonenal; MDA: malondialadehyde; GSH: glutathione.