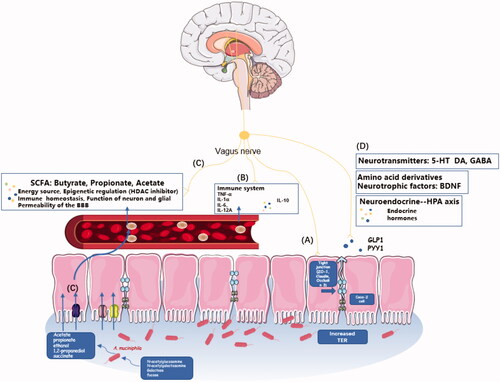
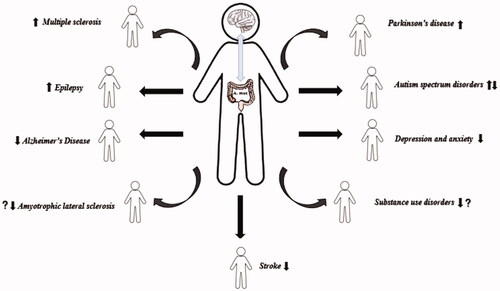
Figures & data
Table 1. Akkermansia muciniphila and its association with different neuropsychiatric diseases.