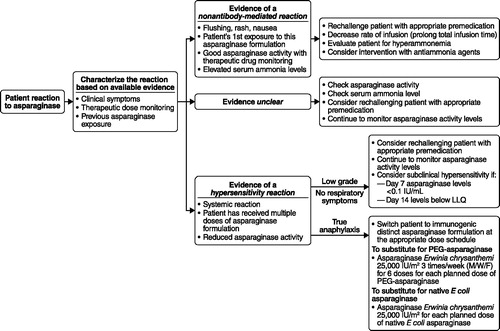
Figures & data
Table 1. Common symptoms of hypersensitivity and infusion reactions by body system.[Citation27]
Table 2. Prevalence of hypersensitivity reactions (any grade) across trials by the method of administration and type of asparaginase.
Table 3. Summary of retrospective comparisons of the prevalence of hypersensitivity reactions with IV and IM PEG-asparaginase.
![Figure 1. Simulation of serum asparaginase activity following repeated administration of intravenous (IV) and intramuscular (IM) asparaginase Erwinia chrysanthemi in two example patients.[Citation17] Reproduced with permission from Albertsen et al. [Citation17].](/cms/asset/5fd01aec-0501-4418-9e21-c7d65eac7072/ilal_a_1213826_f0001_b.jpg)