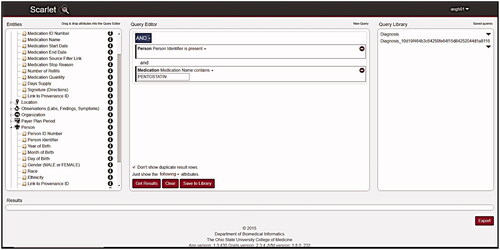
Figures & data
Figure 1. The web-based manual data entry portal (MDE) facilitates entry of information from source documents including pathology reports. The MDE portal can be navigated based on two major tabs divided into patient history (A) and clinical history (B) comprising the entire HCL-PDR data set.
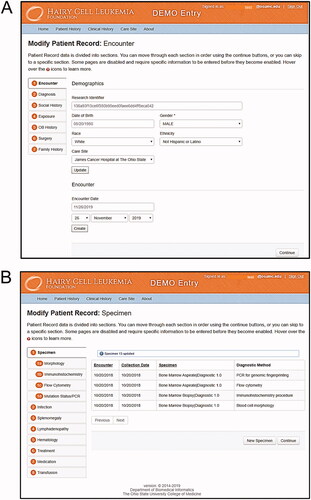
Figure 3. HCL PDR architecture. In this architecture, each institution has a staging database hosted at OSU and has full control of its patients’ data. MDE: manual data entry; ETL: extraction, transfer, and load process.
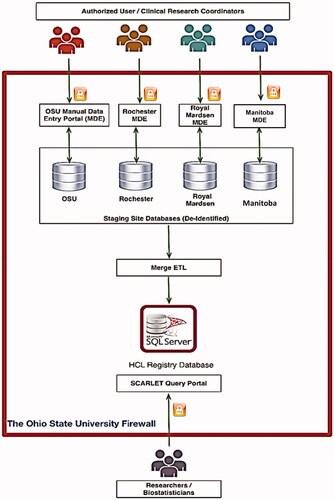
Figure 4. Registry participation. (A) Web-based patients' participation by location (circles) along with the Centers of Excellence (squares). Centers of Excellence that are already part of the registry project have the number of patients enrolled to date indicated next to the representative square. COE: Centers of Excellence; MDA: MD Anderson, Texas, US; MSK: Memorial Sloan Kettering, New York, US; OSU: The Ohio State University, Columbus, US; UR: University of Rochester, Rochester, US; UNM: University of New Mexico, Albuquerque, New Mexico, US; RM: Royal Marsden Hospital, London, UK; USH: University of Southampton, Southampton, UK; PMCC: Peter McCallum Cancer Center, Melbourne, Australia; CCM: Cancer Care Manitoba, Manitoba, Canada. (B) Total number of consented patients at each site. US: patients from United States and outside Center of Excellence, consented via HCLF website.
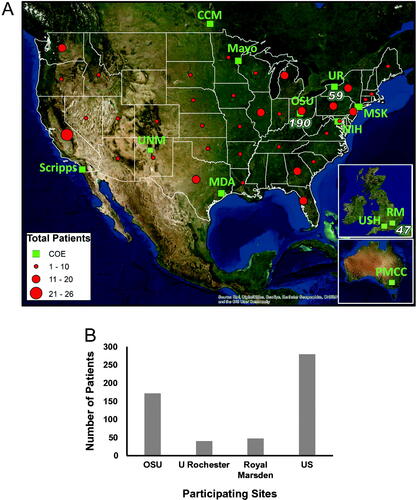
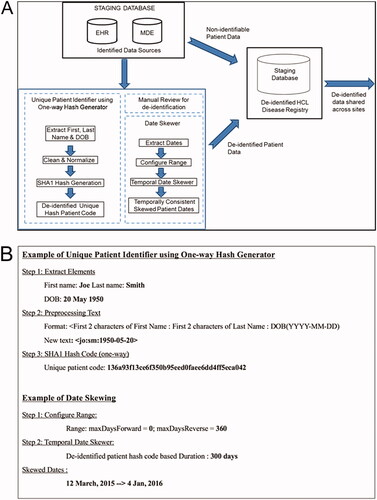
Figure 5. HCL-PDR data de-identification process. A schematic of the de-identification workflow is shown (A). The de-identification process is demonstrated using a sample patient name and date of birth (B). Example of unique patient identifier generator process. EHR: electronic health records; MDE: manual data entry; HCL: hairy cell leukemia; ETL: extraction, transfer, and load process.