Figures & data
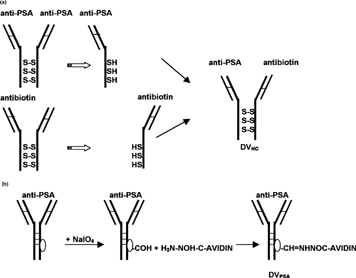
1 Diagram of delivery vehicles preparation. (a) Preparation of bispecific delivery vehicle (DVHc) from anti-PSA and antibiotin monoclonal mouse IgG1: Antibodies were reduced to dissociate and then were allowed to reform SH bonds. (b) Preparation of avidin-conjugated delivery vehicle (DVPSA) from monoclonal mouse IgG1. Carbohydrate residue on the Fc portion of antibodies was oxidized to generate aldehyde, which in turn reacted with avidin-hydrazide.
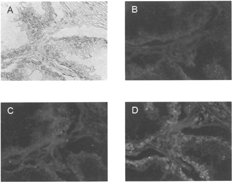
2 Immunoreactivity of heteroconjugate delivery vehicle (DVHc). (A) Tissue sections of paraffin-embedded human prostate carcinoma were treated with unmodified anti-PSA primary antibody followed by streptavidin-conjugated secondary antibody, biotinylated alkaline phosphatase and chromogenic substrate (Fast Red). Staining obtained from these sections served as a reference for PSA expression. (B) DVHC was preincubated with biotinylated HRP and applied to the sections. A three-step amplification system was utilized to detect complex localization as described in the Materials and Methods section of this article. Complexes were visualized with Fast Red. Red staining indicates PSA.
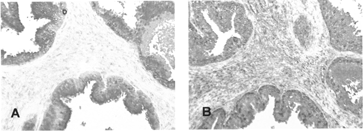
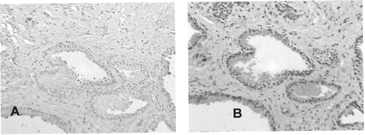
3 Immunoreactivity of avidin-conjugated delivery vehicle (DVPSA). Delivery vehicles (DVIgG or DVPSA) were preincubated with biotinylated HRP and applied to the sections. HRP-DV complexes were detected with chromogenic substrate (AEC): (A) DVIgG, (B) DVPSA.
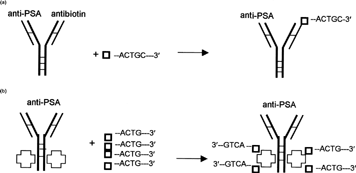
4 Diagram of complex formation between ODN and delivery vehicles. (a) Complex between bispecific delivery vehicle (DVHC) and biotinylated ODN. (b) Complex between avidin conjugated delivery vehicle (DVPSA) and biotinylated ODN (□–ACTG-3′—Biotinylated ODN, —Avidin).
5 Mobility shift assay. Biotinylated ODNs were radioactively labeled with 35S-ddATP and mixed with unlabeled ODN to adjust concentration. A constant amount of ODN (130000 cpm/0.9 nM/binding reaction) was incubated with DVPSA, DVIgG, or DVHC (∼5 μ g). Incubation was carried out at 37°C for 1.5 hr. Twelve percent nondenaturing polyacrylamide gel was run at constant current (100A). The gel was dried and exposed to X-OMAT film (Kodak) for 4 days. As a control ODN alone (Lane 4, Random; [ R], lane 5, EGFR specific) were included in the gel. Lanes 1–3: 35S-Random ODN incubated with DVs. Lanes 6–8: 35S-EGFR-specific ODN incubated with DVs.
![5 Mobility shift assay. Biotinylated ODNs were radioactively labeled with 35S-ddATP and mixed with unlabeled ODN to adjust concentration. A constant amount of ODN (130000 cpm/0.9 nM/binding reaction) was incubated with DVPSA, DVIgG, or DVHC (∼5 μ g). Incubation was carried out at 37°C for 1.5 hr. Twelve percent nondenaturing polyacrylamide gel was run at constant current (100A). The gel was dried and exposed to X-OMAT film (Kodak) for 4 days. As a control ODN alone (Lane 4, Random; [ R], lane 5, EGFR specific) were included in the gel. Lanes 1–3: 35S-Random ODN incubated with DVs. Lanes 6–8: 35S-EGFR-specific ODN incubated with DVs.](/cms/asset/51c7ae95-87ff-457b-8153-d4bf7358db90/idrd_a_16453_f0005_b.gif)