Figures & data
FIG. 1 Mean (± S.D.) amount of nicorandil permeated from carvone-based HPMC gel drug reservoir across rat abdominal skin, EVA 2825 membrane, adhesive-coated EVA 2825, and adhesive-coated EVA 2825-rat skin composite (n=3).
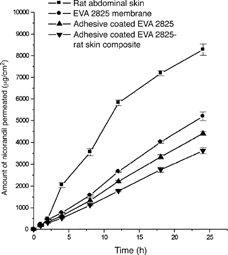
TABLE 1 Mean (±S.D.) in vitro permeation parameters of nicorandil from carvone-based HPMC gel drug reservoir across rat abdominal skin, EVA 2825 membrane, adhesive-coated EVA 2825 membrane, and a composite of adhesive-coated EVA 2825 membrane and rat abdominal skin
FIG. 2 Mean (± S.D.) plasma concentration of nicorandil following the oral administration of immediate release tablet (dose 5 mg) or application of carvone-based TTS (dose 80 mg) in human volunteers (n=6).
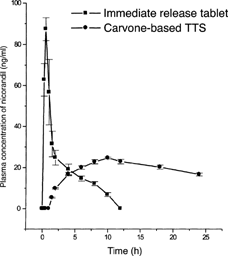
TABLE 2 Mean (±S.D.) pharmacokinetic parameters of nicorandil following oral administration of immediate-release tablet (5 mg) or dermal application of a carvone-based TTS (dose 80 mg) in human volunteers (n=6)