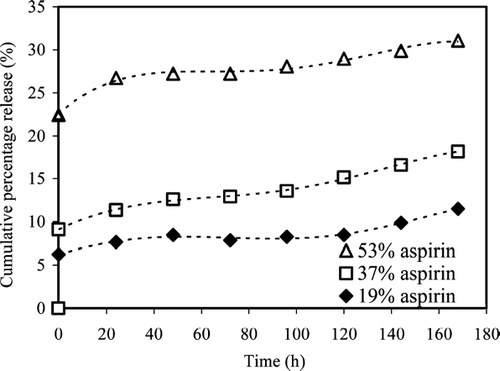
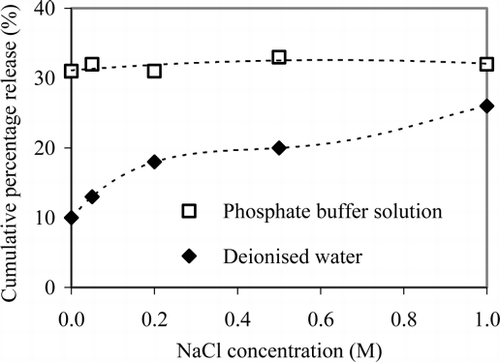
Figures & data
FIG. 1 Schematic representation of the preparation of co-polymer coated magnetic nanocomposite particles using the water replacement method.
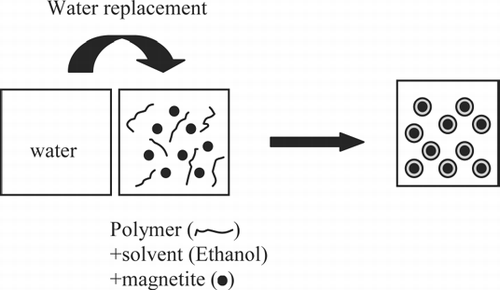
TABLE 1 Ingredients of co-polymer formulation (weight ratio) and freeze-thaw stability ratio of formulation
FIG. 3 Transmission electron microscope images of (a) magnetite and (b) co-polymer-coated magnetic nanocomposite particles.
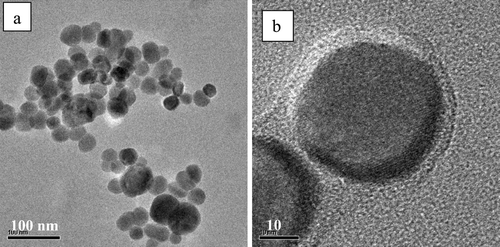
FIG. 4 X-ray powder diffraction patterns of (a) dried polymer powder, (b) magnetite, and (c) polymer-coated magnetic nanocomposite.
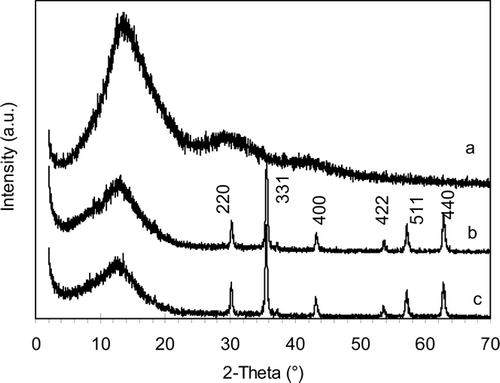
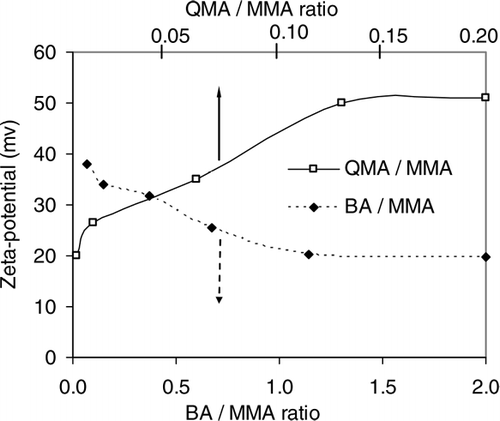
FIG. 5 Zeta potential of (a) co-polymer, (b) magnetite, (c) co-polymer-coated magnetic nanocomposite particles as a function of pH.
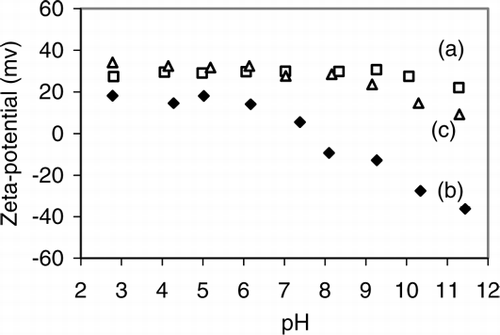
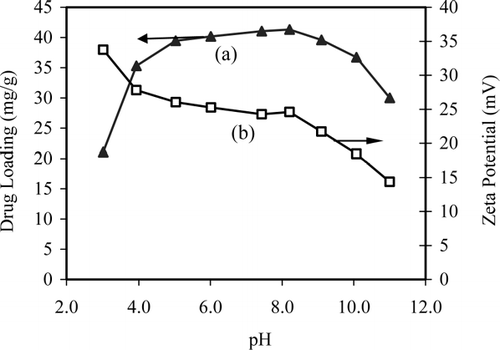
FIG. 6 The effect of pH on (a) aspirin adsorption and (b) zeta potential of magnetic nanocomposite particles (initial drug concentration = 800 mg/L at 25°C).