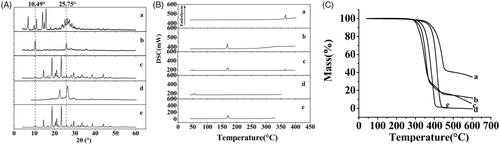
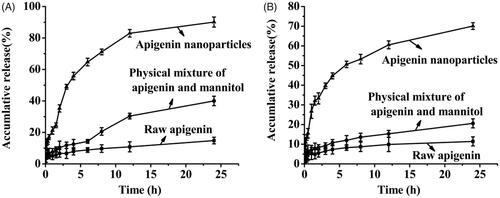
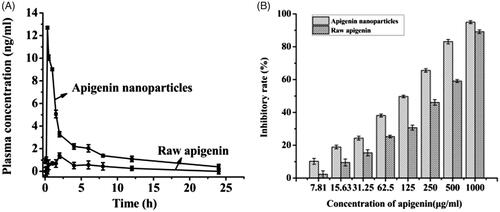
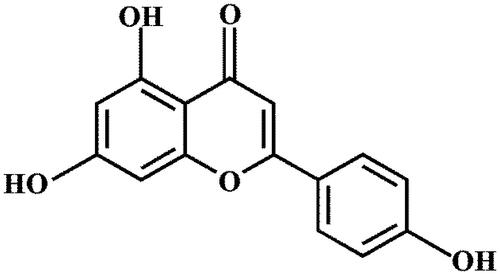
Figures & data
Table 1. Summary of reports available on production of Apigenin particles.