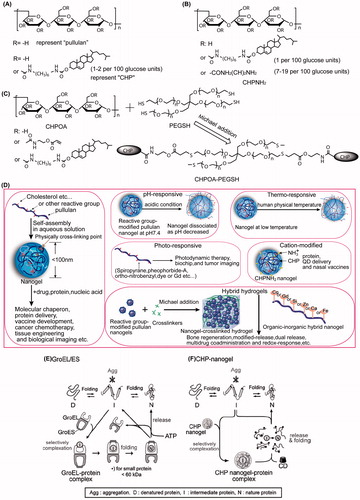
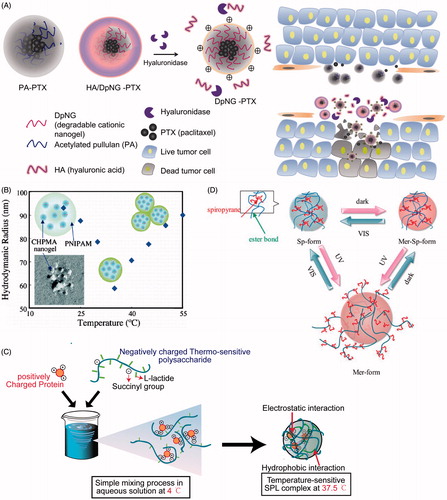
Figures & data
Table 1. Summary of recent studies of CHP nanogels.
Table 2. Summary of recent studies of nanogel crosslinked hydrogels.
Table 3. Summary of recent studies of responsive pullulan nanogels.
Table 4. Clinical studies of CHP nanogels.