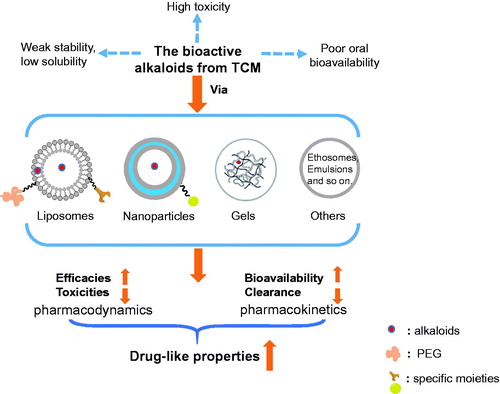
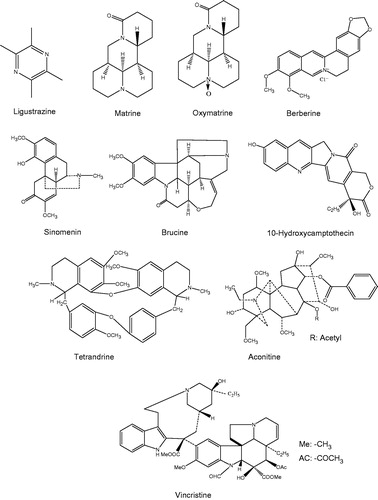
Figures & data
Table 1. Basic information of TCM alkaloids involved in this review.
Table 2. Delivery of alkaloids derived from traditional Chinese medicine by various liposomes.
Table 3. Delivery of alkaloids derived from traditional Chinese medicine by sustained-release delivery systems.
Table 4. Delivery of alkaloids derived from traditional Chinese medicine by transdermal drug delivery systems.