Figures & data
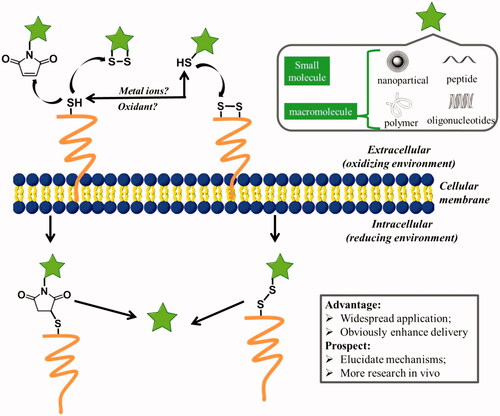
Figure 1. (A) Structures of major membrane lipids (sphingomyelin (SM), phosphatidylcholine (PC), phosphatidylethanolamine (PE) and phosphatidylserine (PS)). (B) A simplified representative illustration of uptake mechanisms through interaction with lipid.
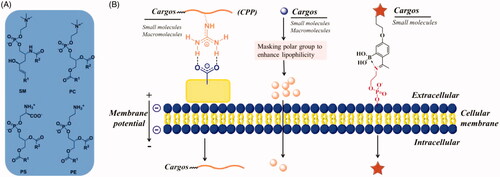
Figure 2. (A) GLUT mediated cellular uptake. (B) Structure-activity relationship of glucose as a substrate for the GLUT1 transporter. (C) The amino acid residues involving the hydrogen-bonding interactions present in the docking model of glucose-platinum conjugate into XylE (PDB 4GBZ). (D) Glucose was introduced onto nanocarriers to enhance retention. Reproduced with permission from ref (Calvaresiet al.,2013). Copyright 2013 Royal Society of Chemistry.
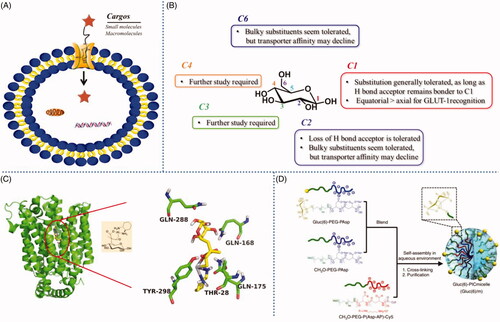
Figure 3. (A) Cartoon depicting liposome cell entry driven by binding interactions with cell surface carbohydrates. (B) Fluorescence image of cells treated with boronic acid modified liposomes. Reproduced with permission from ref (Zhang et al., Citation2018). Copyright Royal Society of Chemistry.
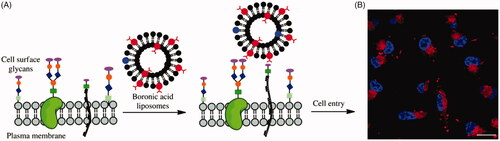
Figure 4. Possible mechanisms for cellular uptake of thiol-reactive groups (including maleimide moiety, disulfide bonds, and free thiols) modified biomolecules (pentacle) upon interaction with exofacial thiols. After these biomolecules were located on the cell surface, they were further internalized to free cargoes through cleavage.