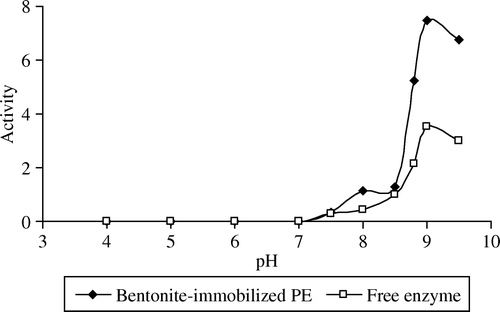
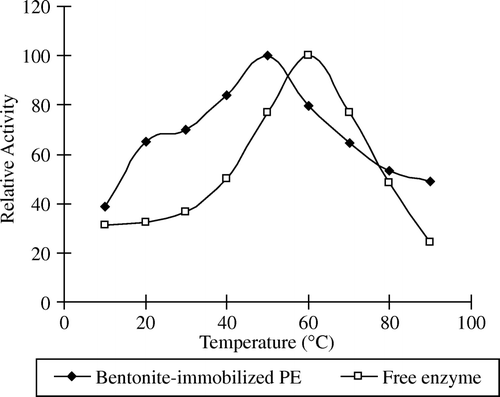
Figures & data
Table 1. Chemical composition of bentonite
Table 2. Purification of pectinesterase enzyme isolated from Malatya apricot
Table 3. Immobilization of PE from Malatya apricot on bentonite
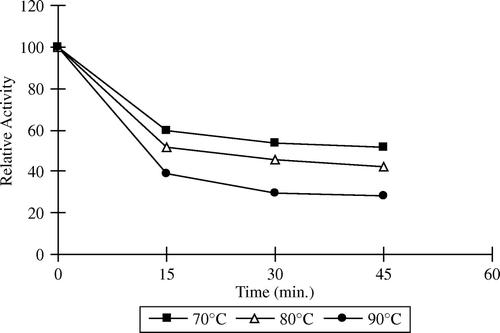
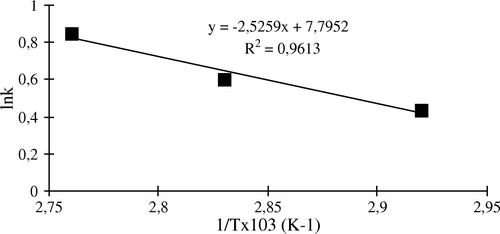
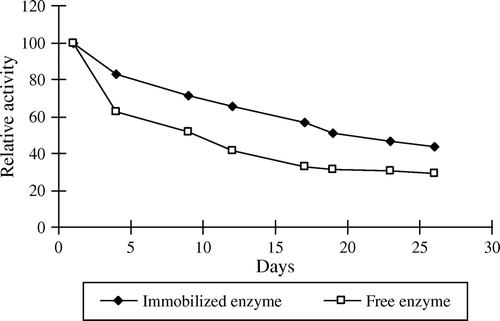
Table 4. Inactivation rate constant and half-lives (t1/2) of free and immobilized apricot PE
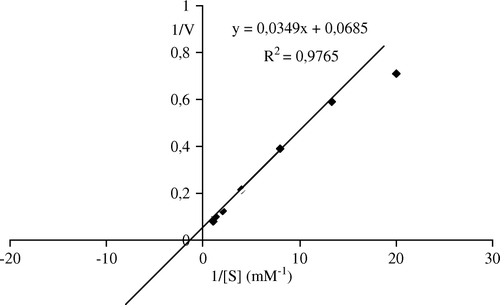
Figure 6. Lineweaver-Burk plots of apricot PE activity as a function of substrate concentration. Reaction mixtures contained varying levels of pectin (0.05–1 mM) in 0.1M NaCl (w/v) and 0.4 ml of partially purified enzyme. The pH of the reaction was 9.0 and the temperature was 30°C.
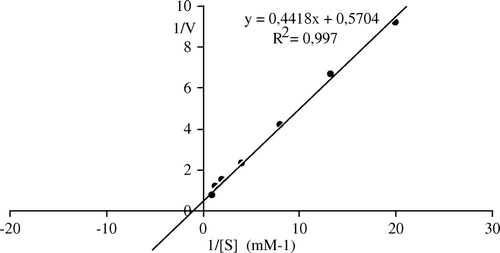
Figure 7. Lineweaver-Burk plots of bentonite-immobilized Malatya apricot PE activity as a function of substrate concentration. Reaction mixtures contained varying levels of pectin (0.05–1 mM) in 0.1M NaCl (w/v) and 0.05 g of patially purified enzyme. The pH of the reaction was 9.0 and the temperature was 30°C.