Figures & data
Table 1. Some available physical and chemical properties of the selected fluorocarbon surfactants
Figure 1. A sketch map showing the formation and absorption of the simulated exhaust gas loaded with toluene: 1, air bottle; 2, rotary flowmeter; 3, valve; 4, vessel 1 (organic exhaust gas producer); 5, thermostatic bath; 6, vessel 2 (organic exhaust gas dilution vessel); 7, gas dispersing device; 8, vessel 3 (glass tube).
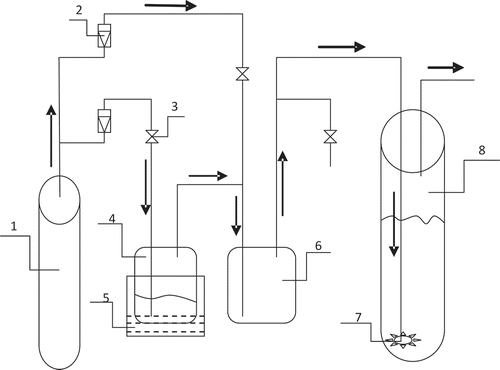
Figure 2. A comparison of saturation toluene concentrations of the absorption solutions of the studied fluorocarbon surfactants with absorbent concentrations of 0.05% and 0.1% obtained by liquid–liquid mass transfer (a) and gas–liquid mass transfer (b).
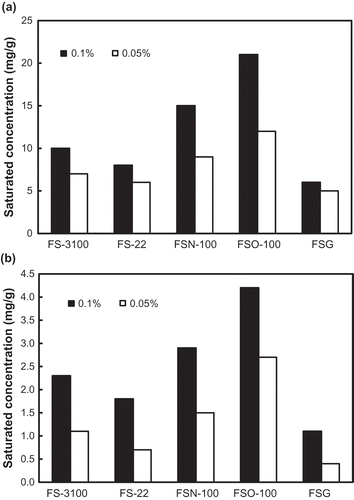
Figure 3. Plot showing the relationship between the two kinds of saturation toluene concentrations obtained respectively by the liquid–liquid mass transfer and gas–liquid mass transfer for absorption solutions of the studied fluorocarbon surfactants.
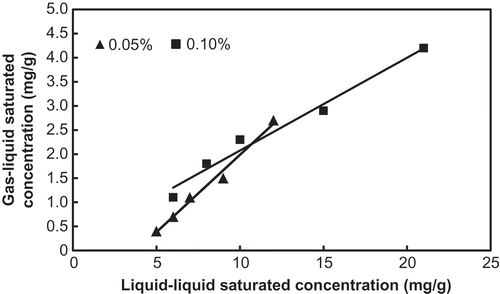
Figure 4. Absorption curves of the absorption solutions of the studied fluorocarbon surfactants to the simulated exhaust gas with an inlet toluene concentration of 3000 mg/m3. The absorbent concentrations in the absorption solutions are 0.05% (a) and 0.1% (b).
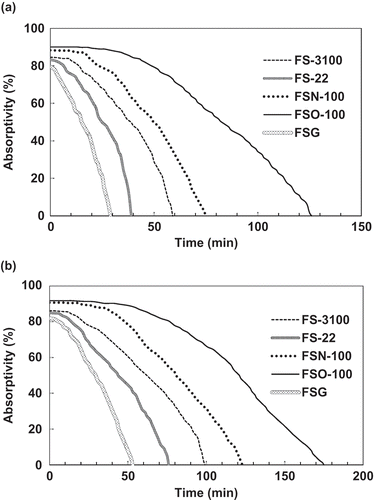
Table 2. Henry’s constant reciprocal values of the absorption solutions with an absorbent concentration of 0.1%
Figure 5. Absorption curves of FSO-100 absorbent solutions with concentrations from 0.01% to 2%. The inlet gas toluene concentration is 3000 mg/m3.
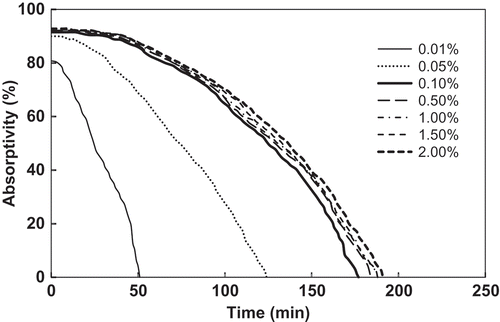
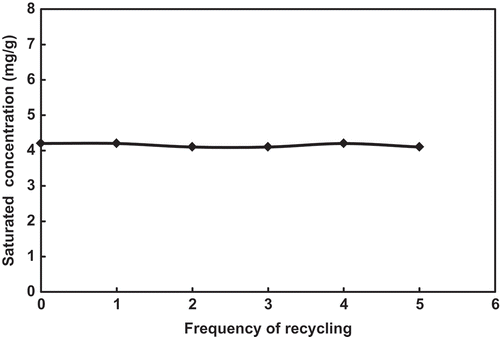
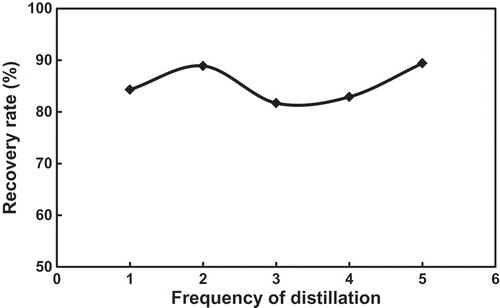
Table 3. Saturation toluene concentrations of the FSO-100 absorbent solutions with different concentrations
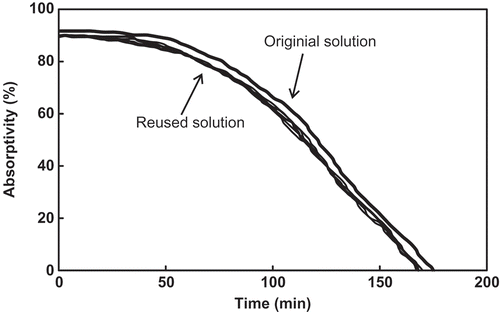
Figure 6. Absorption curves of the FSO-100 absorbent solution to the simulated exhaust gas during reusing.