Figures & data
Figure 1. Mechanical high-energy ball mill Spex 8000 type for an experimental setup to make the particles.
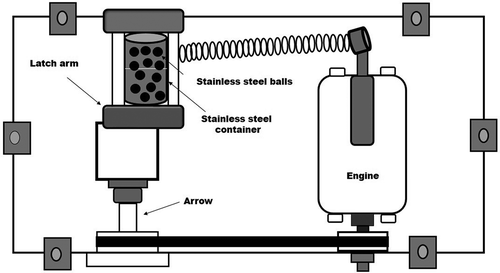
Table 1. Experimental ball milling conditions.
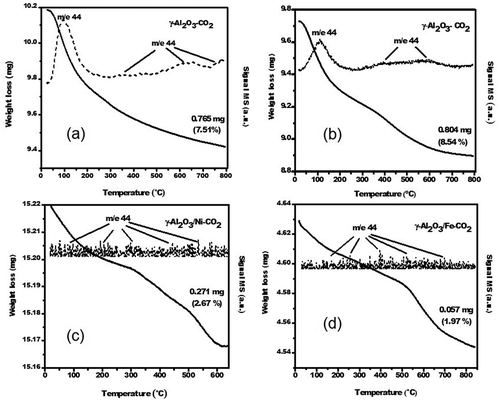
Figure 2. Schematic diagram of the CO2 adsorption on as-prepared porous γ-Al2O3 samples over a Parr 4592 stainless-steel pressure reactor.
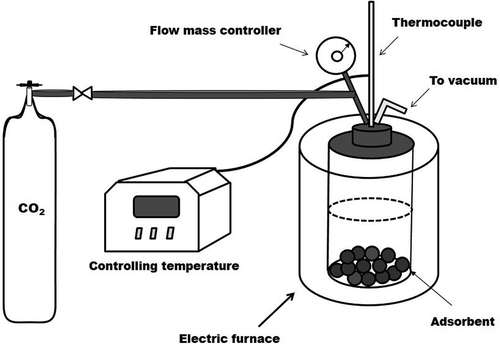
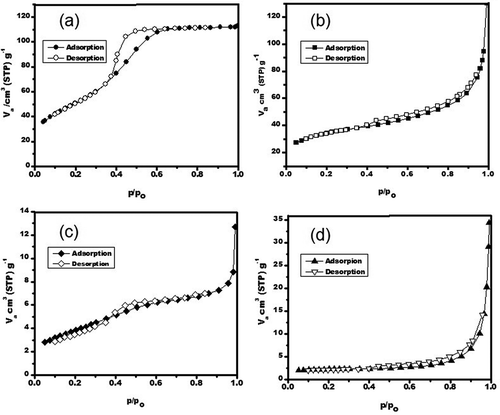
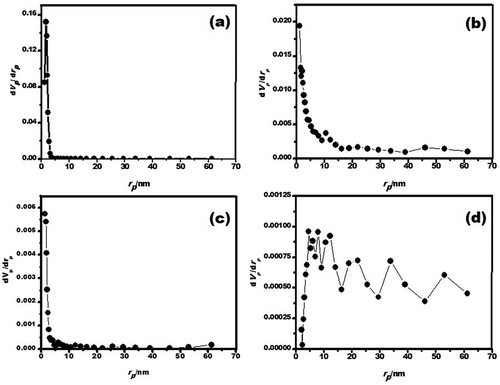
Table 2. Textural properties by nitrogen physisorption of porous γ-Al2O3 samples prepared by solution-combustion and ball-milling processes.
Figure 3. XRD patterns of porous γ-Al2O3 samples prepared by (a) solution combustion, (b) solution combustion after 10 hr of ball milling, (c) γ-Al2O3/Ni, and (d) γ-Al2O3/Fe after 7.5 hr of ball-milling.
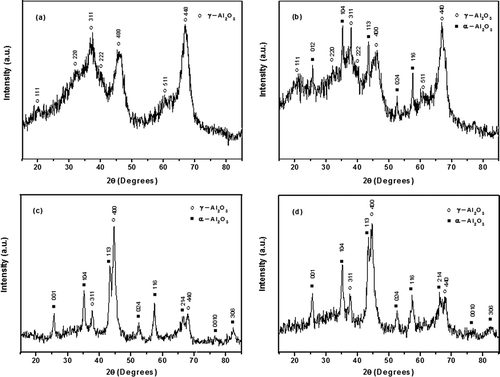
Figure 4. SEM micrographs of porous γ-Al2O3 samples prepared by (a) solution combustion, (b) solution combustion after 10 hr of ball-milling, (c) γ-Al2O3/Ni, and (d) γ-Al2O3/Fe after 7.5 hr of ball-milling.

Table 3. γ-Al2O3 prepared by solution combustion; textural properties and adsorption capacity (qads) as a function of ball milling time.
Figure 5. Nitrogen adsorption–desorption isotherms curves for the porous γ-Al2O3 samples prepared by (a) solution combustion, (b) solution combustion after 10 hr of ball milling, (c) γ-Al2O3/Ni, and (d) γ-Al2O3/Fe after 7.5 hr of ball milling.