Figures & data
Figure 1. Monitoring sheet by organ system in the intensive care unit, and examples of descriptions (partial excerpt). This sheet was created to enable systematic evaluation of drug therapy by organs. It allows for the checking of antimicrobial drug type and dose, bacterial detection, and inflammatory markers related to infection. By sharing monitoring points for each patient among ICU pharmacists, early AS can be implemented. ICU: Intensive care unit, WBC: White blood cell, CRP: C-reactive protein, PCT: Procalcitonin, CD: Clostridioides difficile, VCM: Vancomycin, TDM: Therapeutic Drug Monitoring, TAZ/PIPC: Tazobactam/Piperacillin, MCFG: Micafungin, MRSA: methicillin‐resistant Staphylococcus aureus, F-FLCZ: Fosfluconazole.
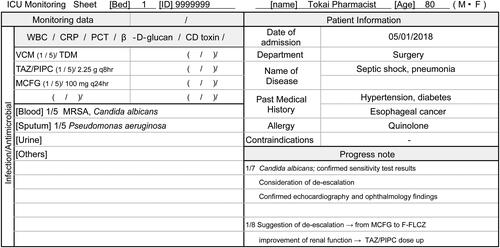
Table 1. Characteristics of patients admitted to the ICU.
Table 2. Number of patients undergoing AS and the number of AS implementations.
Table 3. Types of AS.
Table 4. AUD and DOT of antimicrobial drugs by class.
Table 5. AUD and DOT of broad-spectrum antimicrobial drugs.
Table 6. Bacterial test implementation and resistant bacteria detection.
Table 7. Detection of CD toxins.
Table 8. Thirty-day mortality.
Data availability statement
The data that support the findings of this study are available from the corresponding author, [S.A], upon reasonable request.
