Figures & data
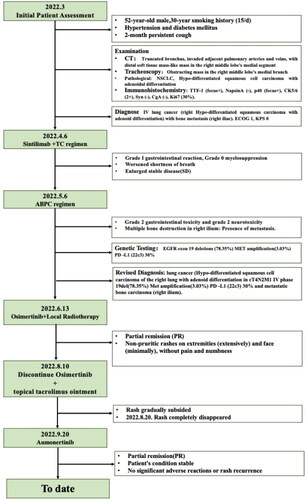
Figure 1. The clinical course according to CT scan findings: (A) (B) March 2022: a truncation observed in the medial segmental bronchus of the right middle lobe, accompanied by a distal mass-like soft tissue lesion; (C) March 2022: Osteolysis of the right iliac bone; (D) (E) April 2022: the constriction in the medial segmental bronchus of the right middle lobe has intensified. Therapeutic assessments indicate a progressive SD; (F) June 2022 chest CT: a reduction in the size of the pulmonary focus in the lung; (G): August 2022 chest CT, two months after starting osimertinib, showed a reduced right middle lobe tumor, evaluated as PR; (H) November 2022: PR observed two months after initiating aumornertinib; (I) PR on aumornertinib in february 2023.
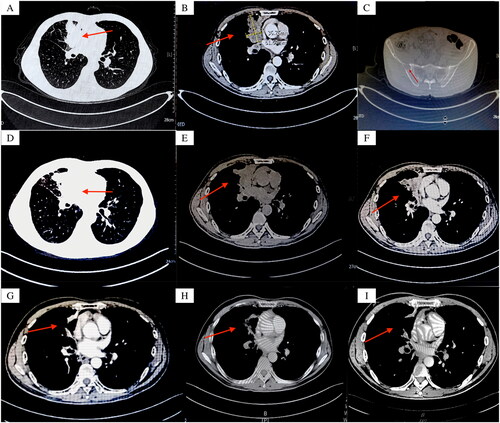
Figure 2. (A) (B) Two months after osimertinib treatment: multiple rashes in the extremities, no itching, pain, and hypoesthesia; (C) (D) (E) (F) Rash gradually subsided.
Table 1. Characteristics of targeted therapy and immunotherapy for skin.