Figures & data
Figure 1 . FTIR spectra of the waste S. fradiae biomass before (a) and after (b) Pb(II) biosorption.
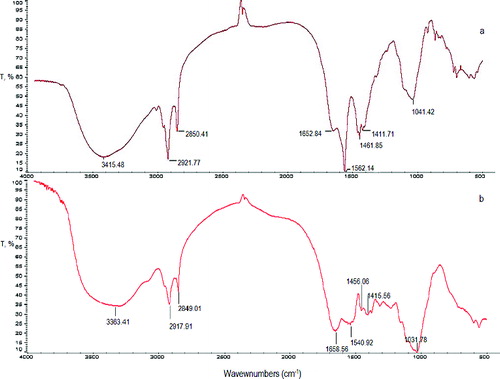
Figure 2. Effect of pH on biosorption of Pb(II) by waste S. fradiae biomass. Note: Ci = 50 mg·dm−3, V = 100 cm3, W = 1 g·dm−3, t = 120 min.
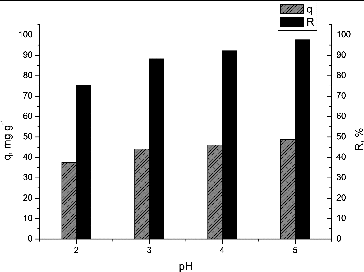
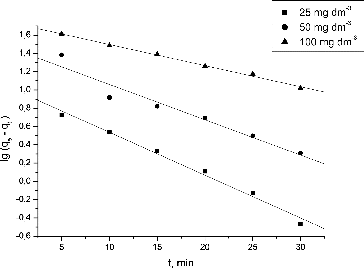
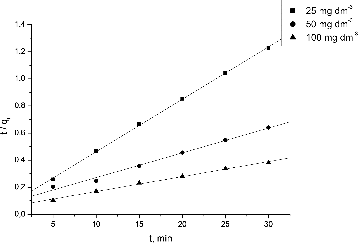
Figure 3. Effect of contact time and initial Pb(II) concentration on the NaOH-pretreated S. fradiae biomass biosorption capacity. Note: pH = 5.0, V = 100 cm3, W = 1 g·dm−3, t = 120 min, Ci = 25, 50, 100 mg·dm−3.
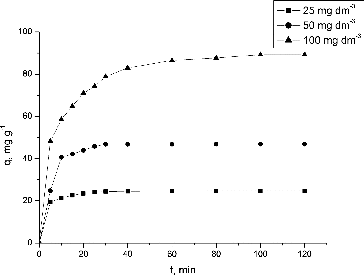
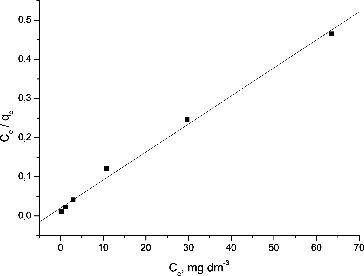
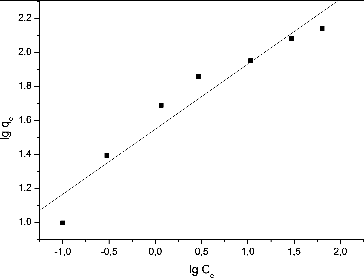
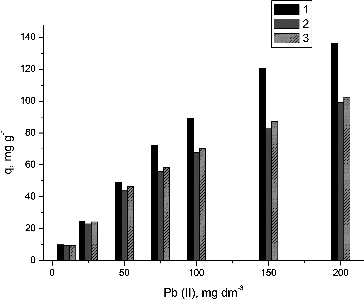
Table 1. Isotherm model constants and correlation coefficients for biosorption of Pb(II) ions.
Table 2. Kinetic constants for Pb(II) biosorption by waste S. fradiae biomass.