Figures & data
Figure 1. Primary cultured neurons in vitro (× 400). Green fluorescence revealed microtubule-associated protein 2 antibody (MAP2) expression; the nuclei were stained blue.
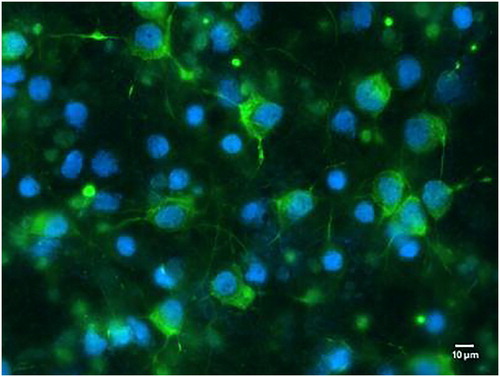
Figure 2. Changes in neuronal cell viability in each group. Compared with control group, NMDA significantly reduced neuronal cell viability. Compared with NMDA group, MK-801(selective NMDA antagonist), rosiglitazone (ROSI, PPAR-γ agonist), and NS398 (selective COX-2 antagonist) both significantly increased cell viability. GW9662 (PPAR-γ antagonist) decreased the cell viability. *P < 0.01, vs. control group; #P < 0.05, ##P < 0.01, vs. NMDA group.
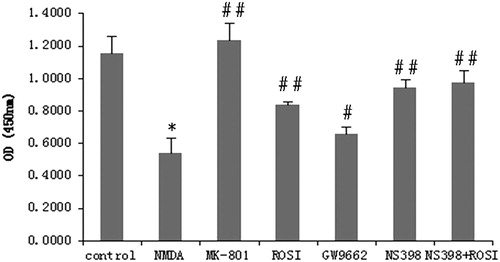
Figure 3. Changes in intracellular Ca2+ in each group. Compared with control group, NMDA significantly increased intracellular Ca2+ concentration. Compared with NMDA group, MK-801(selective NMDA antagonist), rosiglitazone (ROSI, PPAR-γ agonist), and NS398 (selective COX-2 antagonist) both significantly decreased intracellular Ca2+ concentration. GW9662 (PPAR-γ antagonist) increased the cell viability. *P < 0.01, vs. control group; #P < 0.05, ##P < 0.01, vs. NMDA group.
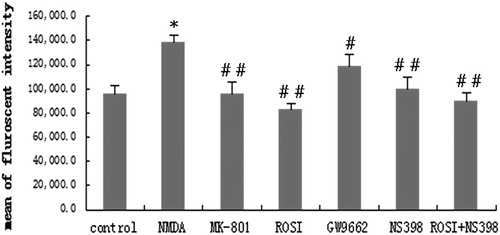
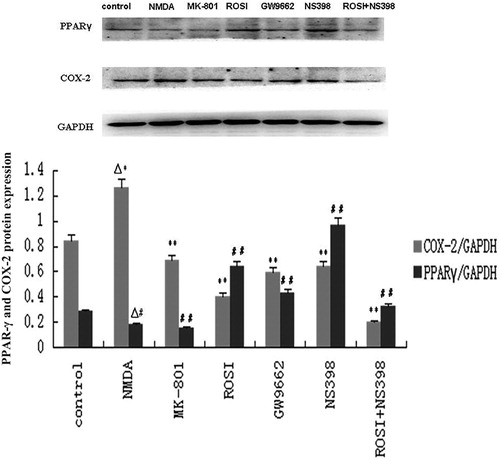
Figure 4. Western blot assay of PPAR-γ and COX-2 protein expression in neurons of each group. Compared with control group, NMDA significantly increased COX-2 protein expression, and decreased PPAR-γ expression. Compared with NMDA group, MK-801(selective NMDA antagonist), rosiglitazone (ROSI, PPAR-γ agonist), and NS398 (selective COX-2 antagonist) both significantly decreased COX-2 protein expression, and increased PPAR-γ expression. GW9662 (PPAR-γ antagonist) increased COX-2 protein expression, and decreased PPAR-γ expression. ▵P < 0.01, vs. control group; **P < 0.01, ##P < 0.01, vs. NMDA group.