Figures & data
Table 1. Effects of different oxygen-vectors on biomass wet weight and enzyme activity.
Figure 1. HPLC of different optical isomers of arginine. Note. The standard sample of dl-arginine was pre-column derivatized. The HPLC column was Supelco chiral column (25 cm × 4.6 mm, 5 μm); with manual injection of 20 μL, UV detection wavelength 254 nm; column temperature 25°; mobile phase A: mobile phase B: 5:1; total flow rate: 1 mL/min.
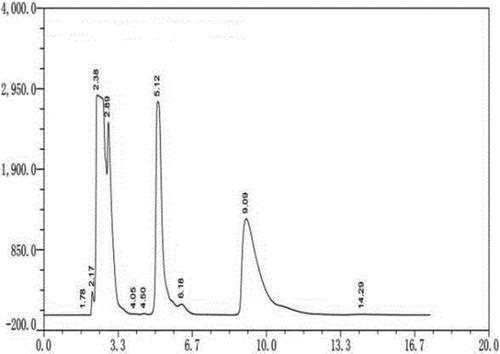
Figure 2. Dynamic changes in dl-arginine content during degradation by Bacillus subtilis HLZ-68. Note. 4 g dl-arginine was added into 50/250 mL initial degradation solution (KH2PO4 5 g/L, Mg2SO4 · 7H2O 2 g/L, CaCl2 0.1 g/L, pH = 7.0) containing 3 g wet biomass.
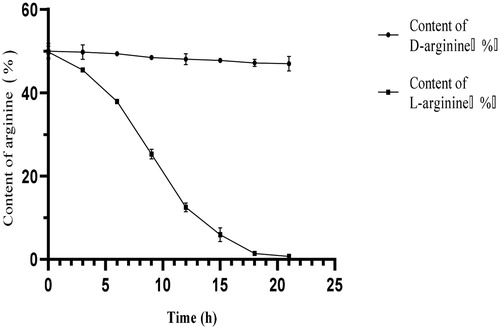
Figure 3. Degradation of dl-arginine in a 5-L fermenter. Note: Bacillus subtilis HLZ-68 cells (100 g of wet weight) were suspended in 2 L of reaction solution containing 180 g of dl-alanine in a jar fermenter. After 9 h of incubation, 90 g of dl-alanine dissolved in small amount of distilled water were added. The reaction was performed at 30 °C with pH 6.0, aeration of 1.0 vvm, and agitation of 400 r/min.
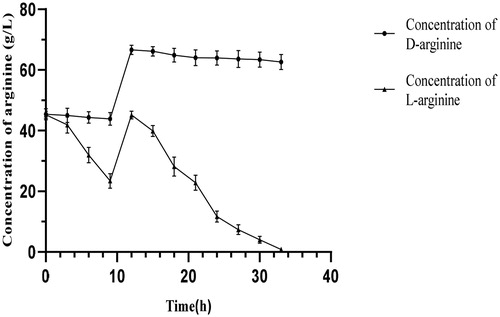
Figure 4. Dynamics of d-arginine adsorption. Note. A strong acid ion exchange resin 732 was used to adsorb d-arginine. The chromatographic column was packed with 150 mL resin. The flow rate of the pump was 2 mL/min. A tube of effluent was collected every 15 min.
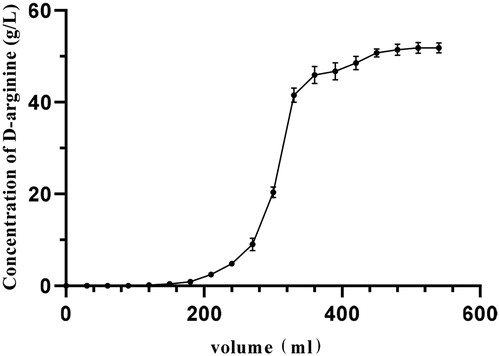
Figure 5. Dynamic elution curve of d-arginine. After the adsorption of d-arginine by ion exchange resin reached a saturation state, d-arginine was eluted. The flow rate of the peristaltic pump was set at 2 mL/min, and the excess impurities in the chromatographic column were washed with 350 mL deionized water; then d-arginine was eluted with 480 mL of 2 mol/L ammonia water at the same flow rate. A tube of eluent was collected every 15 min.
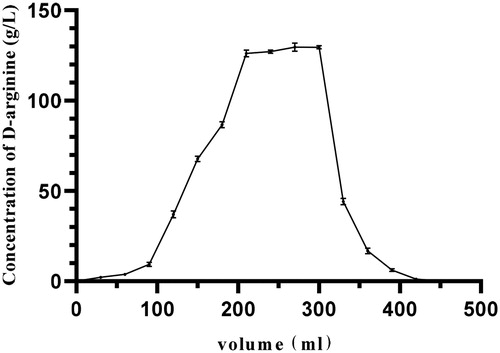
Table 2. Identification of d-arginine.
