Figures & data
Table 1. Monoclonal antibodies authorized in the EU (by October 2020) and included in the PDL in Bulgaria.
Figure 1. Comparison of the authorized mAbs in the EU and the reimbursed ones in Bulgaria by brand name and INN.
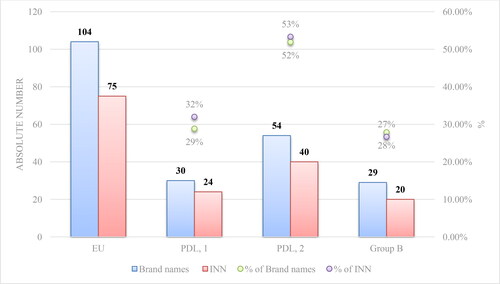
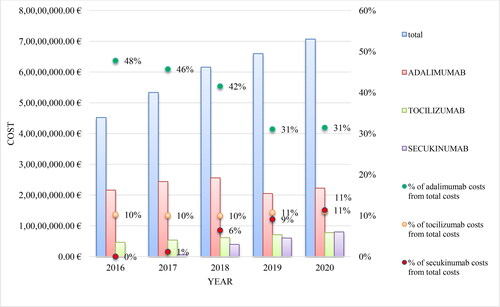
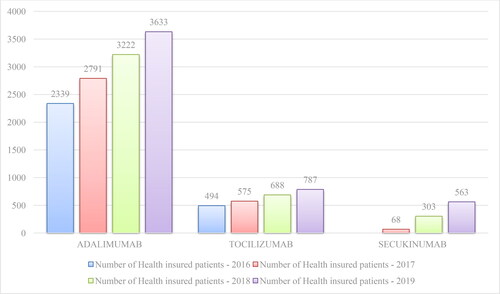
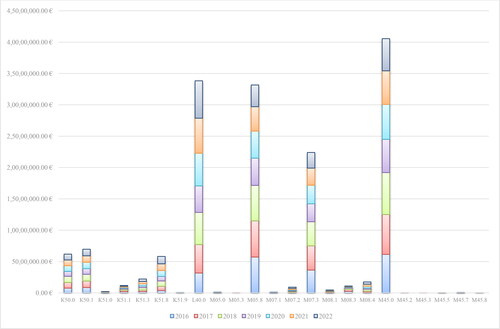
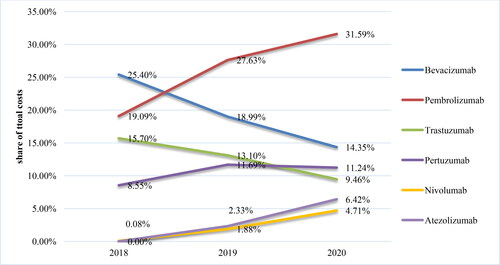
Table 2. Reimbursed costs for mAbs included in PDL, Annex 1 for the period 2016–2020.
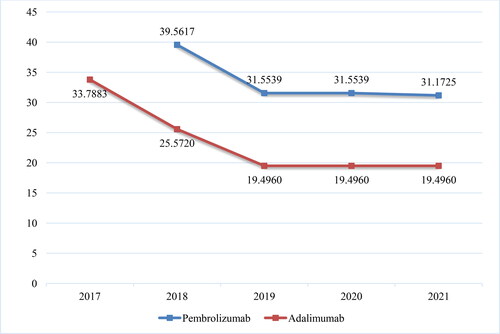
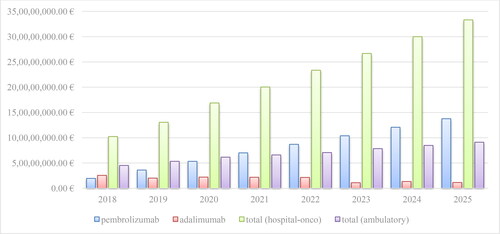
Figure 5. Reimbursed costs for the original product and biosimilars with INN Adalimumab for the period of 2019–2020.
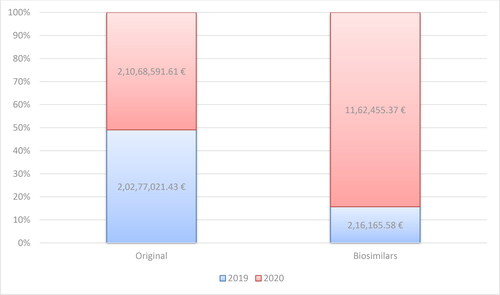
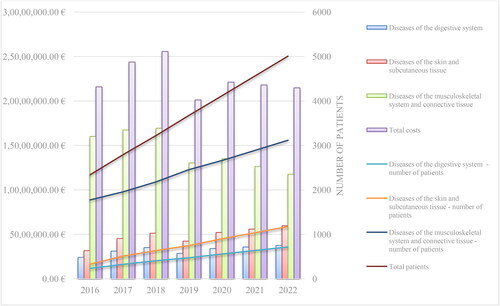
Table 3. mAbs costs from group B*.
Data availability statement
Data sharing not applicable.