Figures & data
Table 1. Demographic data summary.
Table 2. Summary of the countries that participated in the international part of the study.
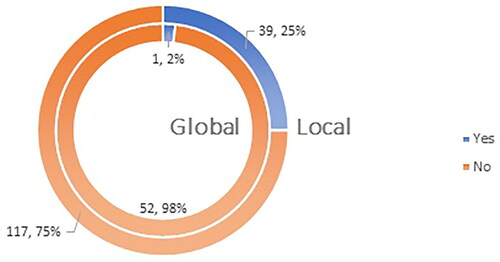
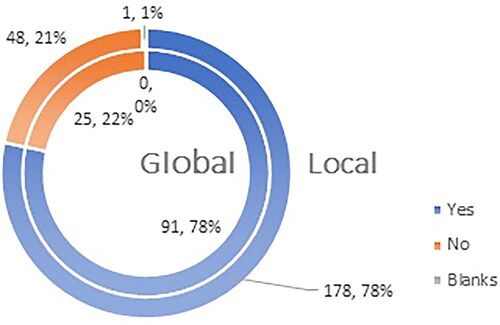
Figure 1. Q1: Do you accept pharmacists to take part in campaigns for increasing population’s awareness concerning the administration of the compulsory immunizations and reimmunizations as per the immunization calendar of your country?
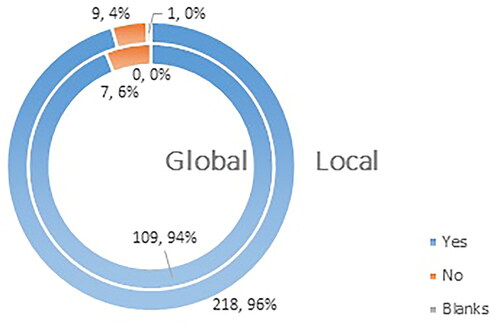
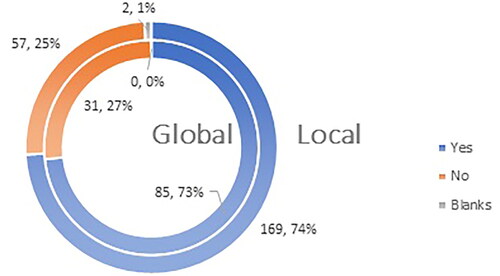
Figure 5. Q5: Are you familiar with the fact that vaccines against COVID-19 have received conditional marketing authorization on the territory of EU for a period of 1 (one) year?
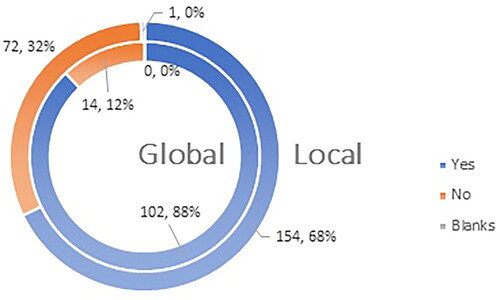
Figure 7. Q12: Would you recommend vaccination to your patients more confidently if vaccines have marketing authorization valid for 5 (five) years?
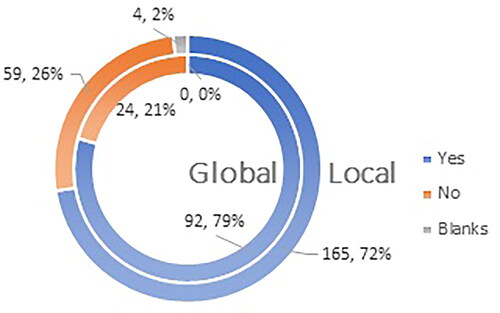
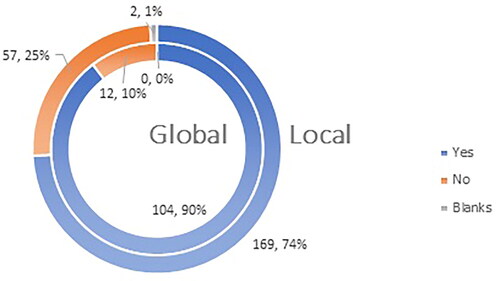
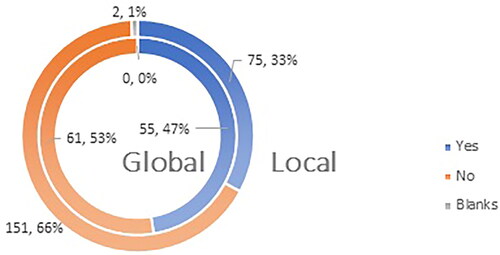
Figure 8. Q11: Do you consider yourself familiar enough with the most frequent ADRs (adverse drug reactions) after administration of a COVID-19 vaccine?
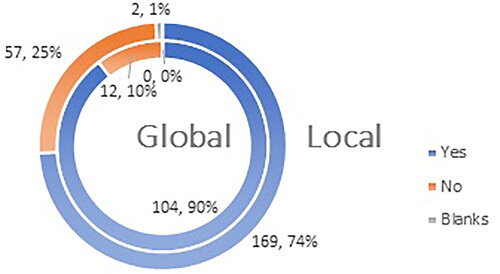
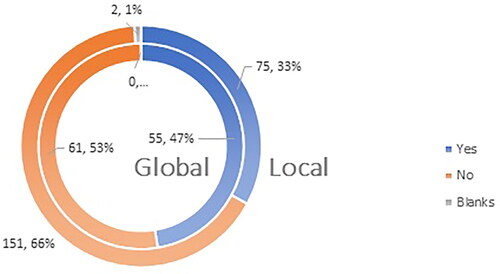
Figure 9. Q16: Do you agree with the statement that the vaccines authorized for use in EU are ‘of high quality, efficacious and safe’?
Data availability statement
The data that support the findings reported in this study are available from the corresponding author [MD] upon reasonable request.