Figures & data
Table 1. Bispecific agents currently tested for psoriasis.
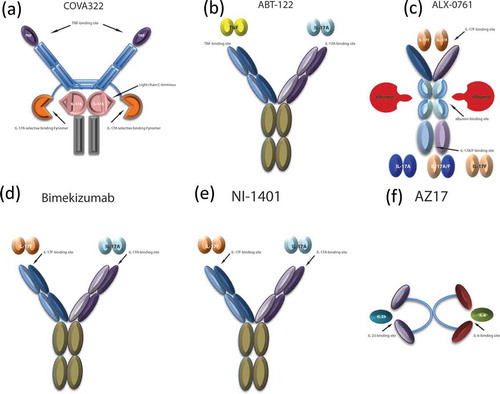
Figure 1. Bispecific therapeutic agents in development for the treatment of psoriasis. (a) COVA322, constituted by a high-affinity IL-17A selective binding Fynomer fused to the C-terminal light chain of the already-marketed fully human anti-TNF-α antibody, adalimumab, maintaining TNF-α binding actvity. (b) ABT-122, a dual variable domain immunoglobulin targeting both TNF-α and IL-17. (c) ALX-0761, a trivalent anti-IL-17A/F nanobody, consisting of an N-terminal IL-17F specific moiety, a C terminal moiety that binds both IL-17A and IL-17F, and a central portion binding albumin. (d) Bimekizumab, a humanized monoclonal antibody inhibiting both IL-17A and IL-17F. (e) NI-1401, a fully human monoclonal antibody neutralizing both IL-17A and IL-17F. (f) AZ17, consisting of two single-chain Fragment variables (scFvs), each one binding IL6 or IL23, and linked by a polyethylene glycol (PEG) moiety.