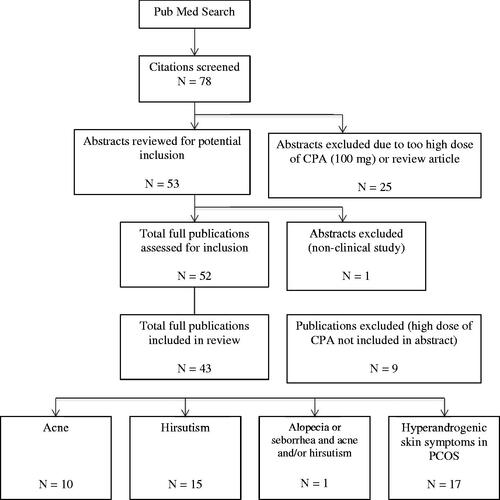
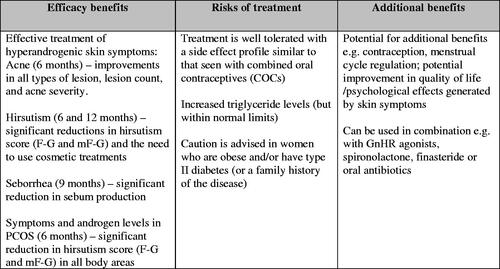
Figures & data
Palombo-Kinne E, Schellschmidt I, Schumacher U, et al. Efficacy of a combined oral contraceptive containing 0.030 mg ethinylestradiol/2 mg dienogest for the treatment of papulopustular acne in comparison with placebo and 0.035 mg ethinylestradiol/2 mg cyproterone acetate. Contraception. 2009;79:282–289. Vartiainen M, de Gezelle H, Broekmeulen CJ. Comparison of the effect on acne with a combiphasic desogestrel-containing oral contraceptive and a preparation containing cyproterone acetate. Eur J Contracept Reprod Health Care. 2001;6:46–53. Carlborg L. Cyproterone acetate versus levonorgestrel combined with ethinyl estradiol in the treatment of acne. Results of a multicenter study. Acta Obstet Gynecol Scand Suppl. 1986;134:29–32. Dieben TO, Vromans L, Theeuwes A, et al. The effects of CTR-24, a biphasic oral contraceptive combination, compared to Diane-35 in women with acne. Contraception. 1994;50:373–382. Charoenvisal C, Thaipisuttikul Y, Pinjaroen S, et al. Effects on acne of two oral contraceptives containing desogestrel and cyproterone acetate. Int J Fertil. 1996;41:423–429. Miller JA, Wojnarowska FT, Dowd PM, et al. Anti-androgen treatment in women with acne: a controlled trial. Br J Dermatol. 1986;114:705–716. Fugere P, Percival-Smith RKL, Cacan SL, et al. Cyproterone acetate/ethinyl estradiol in the treatment of acne. Comparative dose-response study of the estrogen component. Contraception. 1990;40:225–234. Colver GB, Mortimer PS, Dawber RPR. Cyproterone acetate and two doses of oestrogen in female acne; a double-blind comparison. Br J Dermatol. 1988;118:95–99. Carmina E, Lobo RA. Gonadotrophin-releasing hormone agonist therapy for hirsutism is as effective as high dose cyproterone acetate but results in a longer remission. Hum Reprod. 1997;12:663–666. Greenwood R, Brummitt L, Burke B, et al. Acne: double blind clinical and laboratory trial of tetracycline, oestrogen–cyproterone acetate, and combined treatment. Br Med J (Clin Res Ed). 1985;291:1231–1235. Porcile A, Gallardo E. Long-term treatment of hirsutism: desogestrel compared with cyproterone acetate in oral contraceptives. Fertil Steril. 1991;55:877–881. Batukan C, Muderris II, Ozcelik B, Ozturk A. Comparison of two oral contraceptives containing either drospirenone or cyproterone acetate in the treatment of hirsutism. Gynecol Endocrinol. 2007;23:38–44. Ibanez L, Diaz M, Sebastiani G, et al. Treatment of androgen excess in adolescent girls: ethinylestradiol–cyproteroneacetate versus loq-does pioglitazone-flutamide-metformin. J Clin Endocrinol Metab. 2011;96:3361–3366. Belisle S, Love EJ. Clinical efficacy and safety of cyproterone acetate in severe hirsutism: results of a multicentered Canadian study. Fertil Steril. 1986;46:1015–1020. Barth JH, Cherry CA, Wojnarowska F, Dawber RPR. Cyproterone acetate for severe hirsutism: results of a double-blind dose-ranging study. Clin Endocrinol. 1991;35:5–10. Sert M, Tetiker T, Kirim S. Comparison of the efficiency of anti-androgenic regimens consisting of spironolactone, Diane 35, and cyproterone acetate in hirsutism. Acta Med Okayama. 2003;57:73–76. Keleştimur F, Sahin Y. Comparison of Diane 35 and Diane 35 plus spironolactone in the treatment of hirsutism. Fertil Steril. 1998;69:66–69. Sahin Y, Dilber S, Keleştimur F. Comparison of Diane 35 and Diane 35 plus finasteride in the treatment of hirsutism. Fertil Steril. 2001;75:496–500. Taner C, Inal M, Basogul Ö, et al. Comparison of the clinical efficacy and safety of flutamide versus flutamide plus an oral contraceptive in the treatment of hirsutism. Gynecol Obstet Invest. 2002;54:105–108. Tartagni M, Schonauer LM, De Salvia MA, et al. Comparison of Diane 35 and Diane 35 plus finasteride in the treatment of hirsutism. Fertil Steril. 2000;73:718–723. Vegetti W, Testa G, Maggioni P, et al. An open randomized comparative study of an oral contraceptive containing ethinyl estradiol and cyproterone acetate with and without the GnRH analogue goserelin in the long-term treatment of hirsutism. Gynecol Obstet Invest. 1996;41:260–268. Karakurt F. Comparison of the clinical efficacy of flutamide and spironolactone plus ethinyloestradiol/cyproterone acetate in the treatment of hirsutism: a randomised controlled study. Adv Ther. 2008;25:321–328. Falsetti L, Pasinetti E, Ceruti D. Gonadotropin-releasing hormone agonist (GnRH-A) in hirsutism. Acta Eur Fertil. 1994;25:303–306. van Vloten WA, van Haselen CW, van Zuuren EJ, et al. The effect of 2 combined oral contraceptives containing either drospirenone or cyproterone acetate on acne and seborrhea. Cutis. 2002;69:2–15. Bhattacharya SM, Jha A. Comparative study of the therapeutic effects of oral contraceptive pills containing desogestrel, cyproterone acetate, and drospirenone in patients with polycystic ovary syndrome. Fertil Steril. 2012;98:1053–1059. Gokmen O, Senoz S, Gulekli B, et al. Comparison of four different treatment regimes in hirsutism related to polycystic ovary syndrome. Gynecol Endocrinol. 1996;10:249–255. Meyer C, McGrath BP, Teede HJ. Effects of medical therapy on insulin resistance and the cardiovascular system in polycystic ovary syndrome. Diabetes Care. 2007;30:471–478. Kahraman K, Sükür YE, Atabekoğlu CS, et al. Comparison of two oral contraceptive forms containing cyproterone acetate and drospirenone in the treatment of patients with polycystic ovary syndrome: a randomized clinical trial. Arch Gynecol Obstet. 2014;290:321–328. Chung JP, Yiu AK, Chung TK, et al. A randomized crossover study of medroxyprogesterone acetate and Diane-35 in adolescent girls with polycystic ovarian syndrome. J Pediatr Adolesc Gynecol. 2014;27:166–171. Harborne L, Fleming R, Lyall H, et al. Metformin or antiandrogen in the treatment of hirsutism in polycystic ovary syndrome. J Clin Endocrinol Metab. 2003;88:4116–4123. Mastorakos G, Koliopoulos C, Deligeoroglou E, et al. Effects of two forms of combined oral contraceptives on carbohydrate metabolism in adolescents with polycystic ovary syndrome. Fertil Steril. 2006;77:919–927. Creatsas G, Koliopoulos C, Mastorakos G. Combined oral contraceptive treatment of adolescent girls with polycystic ovary syndrome. Lipid profile. Ann N Y Acad Sci. 2000;900:245–252. Luque-Ramírez M, Alvarez-Blasco F, Botella-Carretero JI, et al. Comparison of ethinyl-estradiol plus cyproterone acetate versus metformin effects on classic metabolic cardiovascular risk factors in women with the polycystic ovary syndrome. J Clin Endocrinol Metab. 2007;92:2453–2461. Erkkola R, Hirvonen E, Luikku J, et al. Ovulation inhibitors containing cyproterone acetate or desogestrel in the treatment of hyperandrogenic symptoms. Acta Obstet Gynecol Scand. 1990;69:61–65. Morin-Papunen LC, Vauhkonen I, Koivunen RM, et al. Endocrine and metabolic effects of metformin versus ethinyl estradiol-cyproterone acetate in obese women with polycystic ovary syndrome: a randomized study. J Clin Endocrinol Metab. 2000;85:3161–3168. Dahlgren E, Landin K, Krotkiewski M, et al. Effects of two antiandrogen treatments on hirsutism and insulin sensitivity in women with polycystic ovary syndrome. Hum Reprod. 1998;13:2706–2711. Lemay A, Dodin S, Turcot L, et al. Rosiglitazone and ethinyl estradiol/cyproterone acetate as single and combined treatment of overweight women with polycystic ovary syndrome and insulin resistance. Hum Reprod. 2006;21:121–128. Couzinet B, Le Strat N, Brailly S, Schaison G. Comparative effects of cyproterone acetate or a long-acting gonadotropin-releasing hormone agonist in polycystic ovarian disease. J Clin Endocrinol Metab. 1986;63:1031–1035. Acién P, Mauri M, Gutierrez M. Clinical and hormonal effects of the combination gonadotrophin-releasing hormone agonist plus oral contraceptive pills containing ethinyl-oestradiol (EE) and cyproterone acetate (CPA) versus the EE-CPA pill alone on polycystic ovarian disease-related hyperandrogenisms. Hum Reprod. 1997;12:423–429. De Leo V, Fulghesu AM, la Marca A, et al. Hormonal and clinical effects of GnRH agonist alone, or in combination with a combined oral contraceptive or flutamide in women with severe hirsutism. Gynecol Endocrinol. 2000;14:411–416. Golland IM, Elstein ME. Results of an open one-year study with Diane-35 in women with polycystic ovarian syndrome. Ann N Y Acad Sci. 1993;687:263–271.  https://orcid.org/0000-0002-8486-7861
https://orcid.org/0000-0002-8486-7861