Figures & data
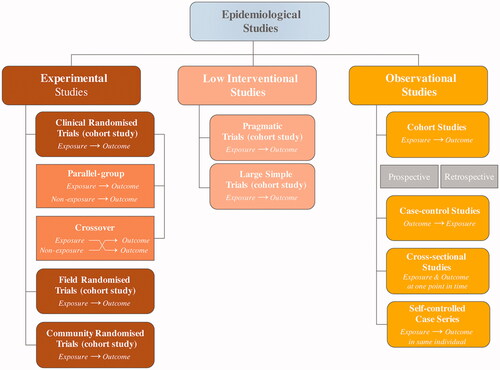
Table 1. Strengths and weaknesses of different types of randomised clinical trials and observational studies [Citation3,Citation5–8].
Table 2. CEBM grades of recommendation.
Table 3. Circumstances under which observational studies are valuable and can be supportive of randomised clinical trials.
Table 4. Limitations of randomised clinical trials versus observational studies.
Figure 3. Causal diagram of factors confounding the relationship between maternal obesity and birth defects. Adapted from Hernán et al. [Citation38].
![Figure 3. Causal diagram of factors confounding the relationship between maternal obesity and birth defects. Adapted from Hernán et al. [Citation38].](/cms/asset/1ba2ea18-b050-404d-80e8-93b42737436b/iejc_a_1361528_f0003_c.jpg)
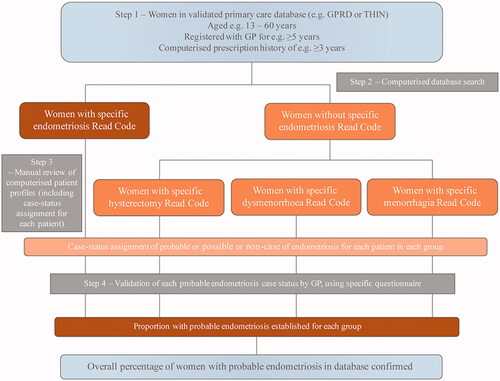
Figure 4. Flowchart exemplifying typical steps for ascertainment and case validation when using a primary care database in an observational study. GP: general practitioner; GPRD: General Practice Research Database; THIN: The Health Improvement Network.