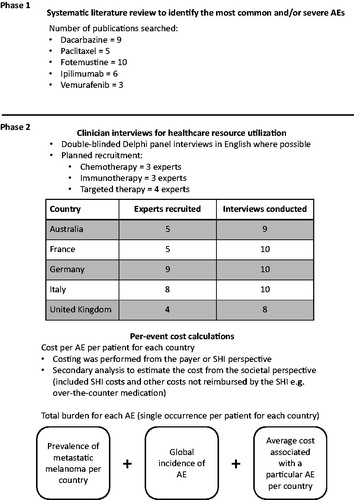
Figures & data
Table 1. Epidemiological and treatment usage data from 5 countries.
Table 2. List of the most common (>20% any grade or >5% grade 3 or higher) adverse events associated with chemotherapy (dacarbazine, paclitaxel and fotemustine), immunotherapy (ipilimumab), and targeted therapy (vemurafenib).
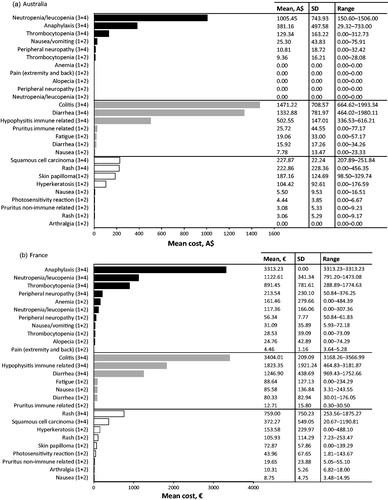
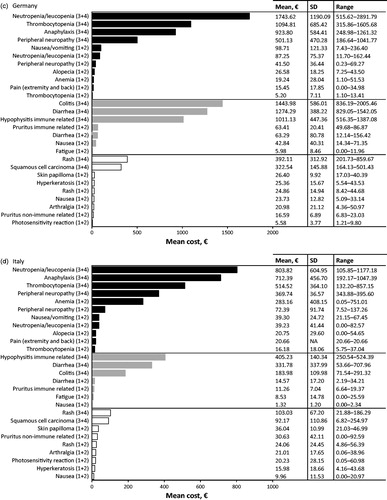
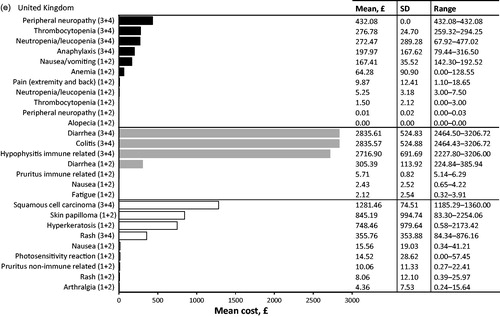
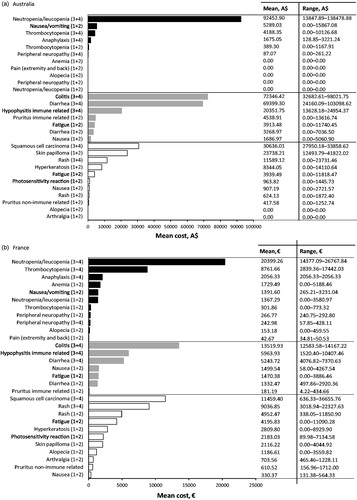
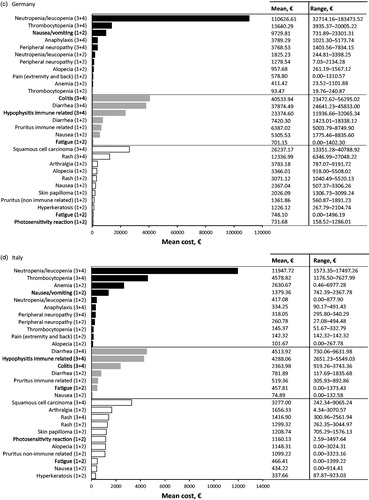
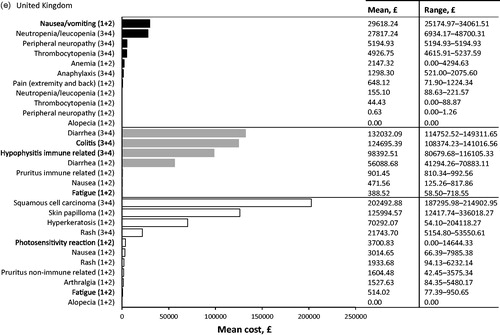
Table 3. Top 10 most cost intensive adverse events (AE) (based on per event per patient) from the statutory health insurance perspective. Adverse events due to chemotherapy (dark grey), immunotherapy (light grey), and targeted therapy (white) are ranked from most expensive to least expensive. All AEs listed are grade 3 or 4 unless specified.