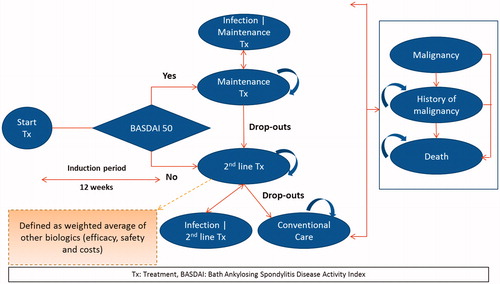
Figures & data
Table 1. Drug acquisition costs for biologics for AS patients in Canada.
Table 2. Summary of ICER results for secukinumab vs biologics by varying base-case assumptions.
Table 3. Total costs, QALYs for biologic-naïve AS patients over a lifetime horizon (60 years).
Table 4. Costs and QALYs breakdown.
Figure 2. Probability of net monetary benefit of AS treatments at different willingness-to-pay thresholds. NMB, Net monetary benefit; SEC, secukinumab; ADA, adalimumab; CER P, certolizumab pegol; ETN, etanercept; GOL, golimumab; INF, infliximab.
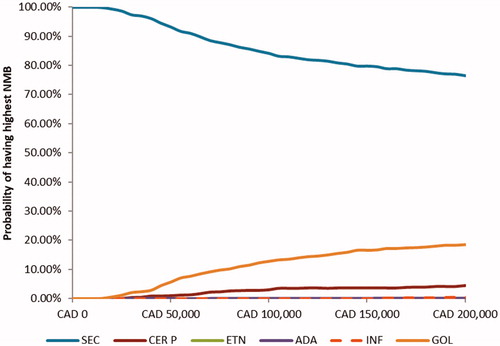
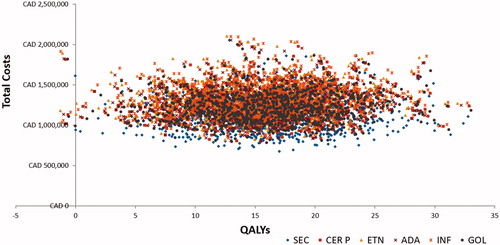
Figure 3. Cost-effectiveness scatter plot. ADA, adalimumab; CAD, Canadian dollar; CE, cost-effectiveness; CER P, certolizumab pegol; ETN, etanercept; GOL, golimumab; INF, infliximab; SEC, secukinumab; QALYs, quality adjusted life years.