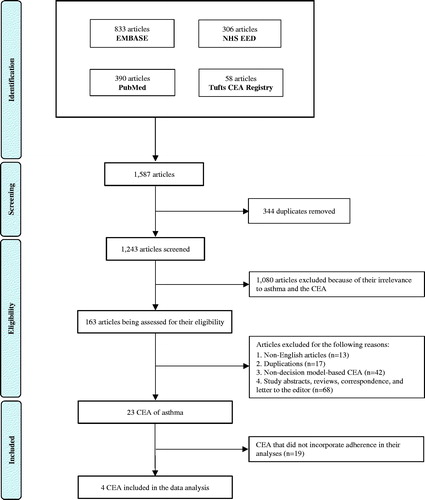
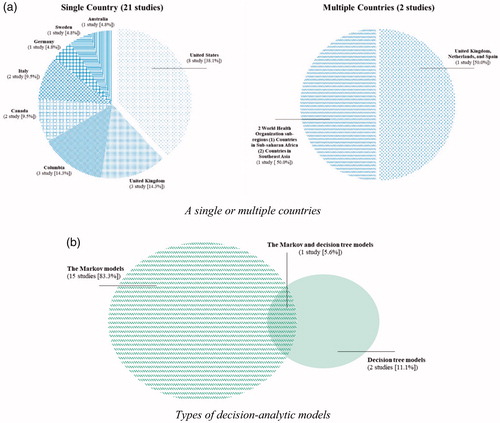
Figures & data
Table 1. Characteristics of the included studies.
Table 3. Quality assessment of the study methodology.
Table 4. Quality assessment of the reported studies.