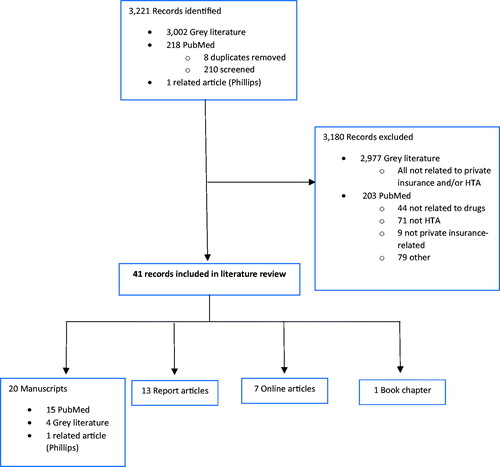
Figures & data
Table 1. Summary of 20 identified MEDLINE manuscripts for the literature review.
Table 2. Important private payer value assessment parameters identified from expert panel.
Table 3. Applicability of public HTA analytic approaches and value parameters for private payer decision-making.