Figures & data
Figure 1. Study design. Abbreviation. AF, atrial fibrillation. The identification period in the figure is shown as an example of case selection for the dabigatran vs. warfarin comparison.
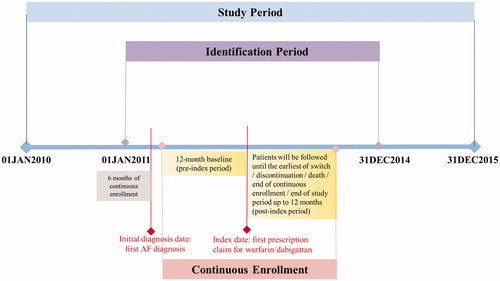
Table 1. Sample size before and after propensity score matching.
Figure 2. Patient attrition. Abbreviations. AF, atrial fibrillation; NOAC, novel oral anticoagulant; NVAF, non-valvular atrial fibrillation; OAC, oral anticoagulant.
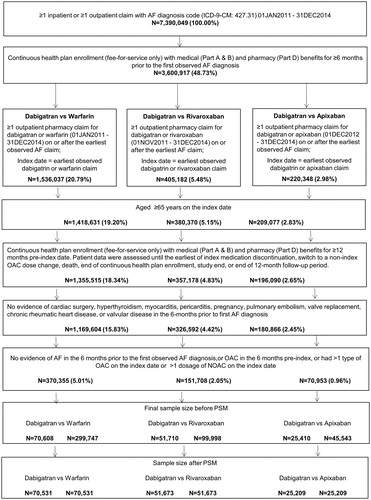
Table 2. Baseline demographics and clinical characteristics (before propensity score matching).
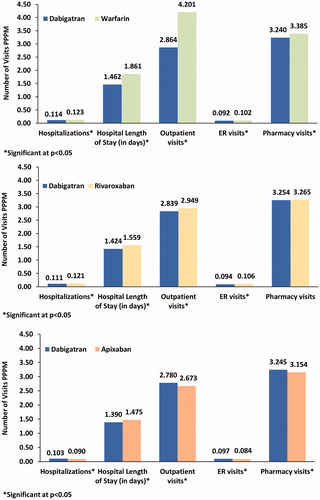
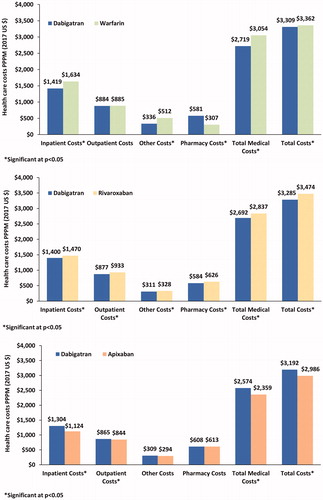
Table 3. Baseline demographic, clinical characteristics, costs & HRU of newly-diagnosed and newly-treated NVAF patients after PSM.
Supplemental data for this article is available online at https://doi.org/10.1080/13696998.2019.1672698.
Download MS Excel (72.8 KB)Data availability
The raw insurance claims data used for this study originate from Medicare data, which are available from the Centers for Medicare and Medicaid through ResDAC (https://www.resdac.org/).