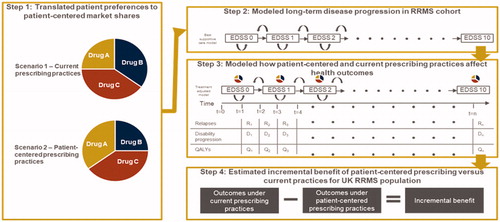
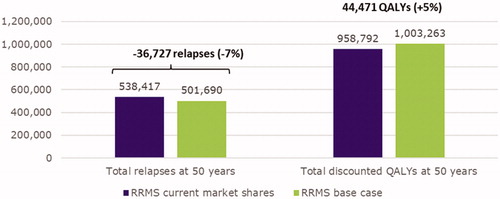
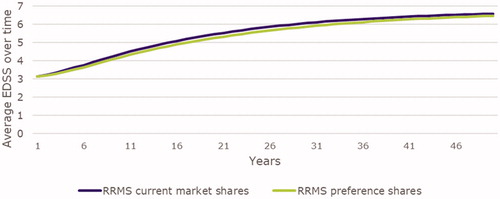
Figures & data
Table 1. Key inputs of the economic model.
Table 2. Preference share estimation results.