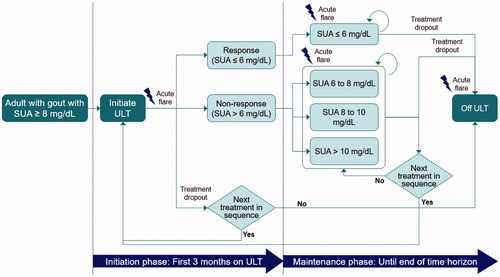
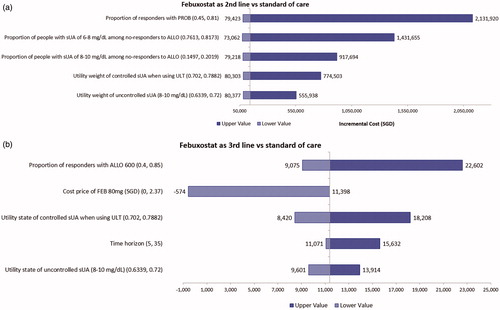
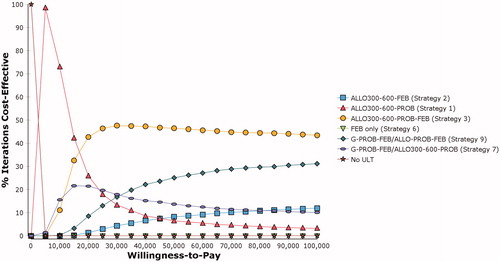
Figures & data
Table 1. Treatment sequences included in the model.
Table 2. Model inputs: clinical parameters.
Table 3. Model inputs: cost parameters.
Table 4. Results from the base-case analysis (SGD).