Figures & data
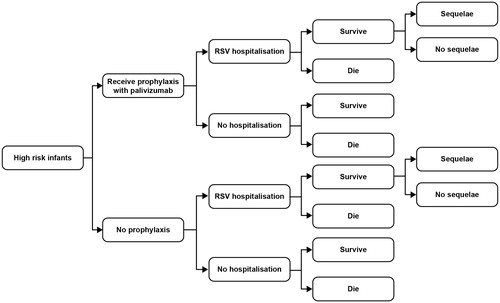
Figure 1. High-risk patient populations endorsed by JCVI for palivizumab in existing guidance.Citation3

Table 1. Clinical and cost inputs of original Bentley et al model and the updated model.
Table 2. Predicted dose of palivizumab for babies born <29 wGA, aged <6 months of age at the onset of the RSV season (October) and discharged at 36 weeks.
Table 3. Summary of results per hundred infants from the base case model.
Table 4. Summary of results per hundred infants, scenario five doses of palivizumab, costs of respiratory sequelae for 2 years and a decrease in utility due to respiratory sequelae for 5 years.
Table 5. Summary of results per hundred infants, scenario respiratory sequalae (costs and utilities) continue until age 7 years, 3.7 doses of palivizumab.
Table 6. Probability that palivizumab is cost-effective for different CE thresholds.