Figures & data
Figure 1. Calculator structure. Abbreviations. N/A, not applicable; SOC, standard of care. *It was assumed that the use of SOC drugs would not be affected by the concomitant use of lenzilumab, resulting in no cost differences for SOC drugs between both groups. This assumption was supported by the balanced use of remdesivir and corticosteroids in both treatment arms of the LIVE-AIR trialCitation15. **Rehospitalization costs are optional in the calculator. They were not included in the base case analysis but were included for scenario analysis #5.
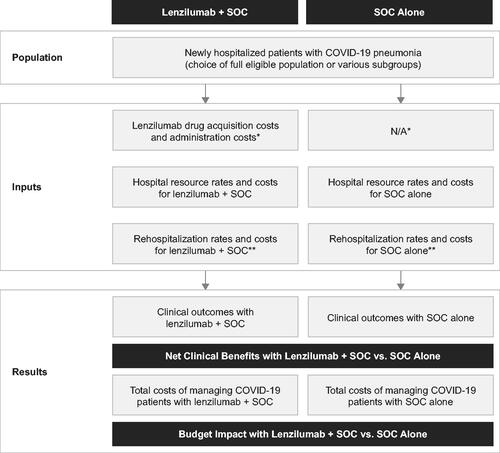
Table 1. Treatment efficacy inputs from LIVE-AIR trial dataCitation34a.
Table 2. Hospital resource costs and use inputs from LIVE-AIR trial dataa.
Table 3. Estimated clinical benefits of lenzilumab plus SOC over SOC alone per treated patient.
Table 4. The estimated economic impact of lenzilumab plus SOC versus SOC alone per treated patient.
Table 5. Sensitivity analyses.
Supplemental Material
Download MS Word (54.1 KB)Data availability statement
The data that support the findings of this study are available through the link below: https://doi.org/10.1101/2021.05.01.21256470
