Figures & data
Figure 1. Model diagram.
Abbreviations. 1L, first-line; NGS, next-generation sequencing; PCR, polymerase chain reaction.
Notes:
1. The estimated costs associated with delaying treatment were calculated as the time to initiation of appropriate targeted therapy times the estimated weekly cost during the pre-diagnosis phase.
2. The estimated costs associated with the initiation of treatment (i.e. immunotherapy and/or chemotherapy) prior to receiving test results were calculated as the time to initiation of appropriate targeted therapy times the weekly drug and medical costs associated with the treatment regimen.
3. Appropriate therapy was defined as targeted therapy for those with an actionable mutation or non-targeted therapy (i.e. immunotherapy and/or chemotherapy) for those without an actionable mutation. Patients who initiated immunotherapy and/or chemotherapy (i.e. non-targeted therapy) prior to receiving test results who were negative for tested alterations continued to receive non-targeted therapy.
4. Wasted expenditures associated with initiating suboptimal treatment (i.e. immunotherapy and/or chemotherapy) were calculated as the estimated incremental medical costs associated with targeted therapy as second line versus first line, times the average duration for the line of therapy.
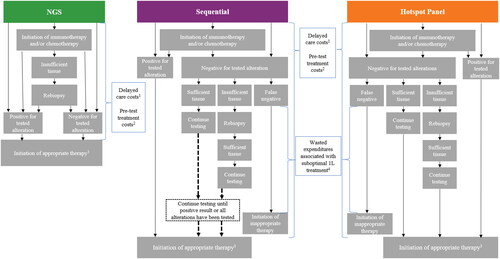
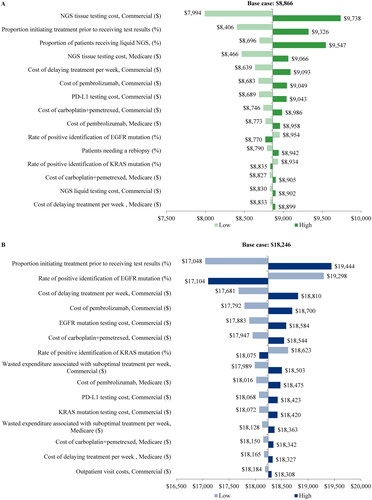
Table 1. Clinical output summary.
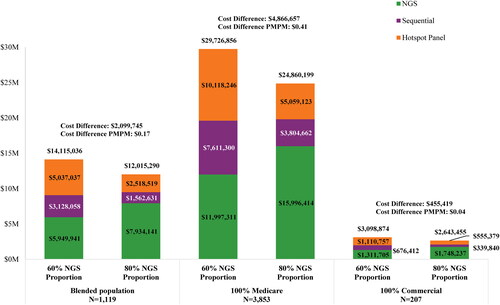
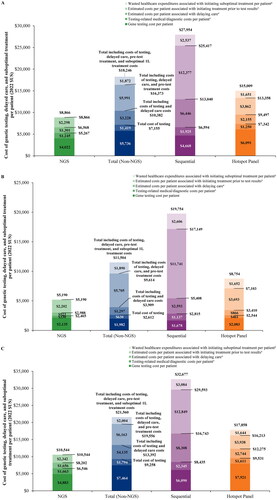
Figure 2. Cost of genetic testing, delayed care, and suboptimal treatment, per testing strategy, among the (A) blended population,1 (B) Medicare only population, and (C) commercial only population.
Abbreviations. 1L, first-line; NGS, next-generation sequencing; US, United States.
Notes:
1. The blended population assumed that 25% of members had Medicare coverage and 75% of members had commercial coverage.
2. Wasted expenditures associated with initiating suboptimal treatment (i.e. immunotherapy and/or chemotherapy) were calculated as the estimated incremental medical costs associated with targeted therapy as second line versus first line, times the average duration for the line of therapy.
3. The estimated costs associated with the initiation of treatment (i.e. immunotherapy and/or chemotherapy) prior to receiving test results were calculated as the time to initiation of appropriate targeted therapy times the weekly drug and medical costs associated with the treatment regimen.
4. The estimated costs associated with delaying treatment were calculated as the time to initiation of appropriate targeted therapy times the estimated weekly cost during the pre-diagnosis phase.
5. Costs include rebiopsy, outpatient/specialist visits, and PD-L1 testing costs.
Supplemental Material
Download MS Word (103 KB)Data availability statement
The data underlying this article are available in the article and in its online supplementary materials.