Figures & data
Figure 1. Patient disposition.
ERA, endothelin receptor antagonist; PAH, pulmonary arterial hypertension; PBS, Pharmaceuticals Benefits Scheme.
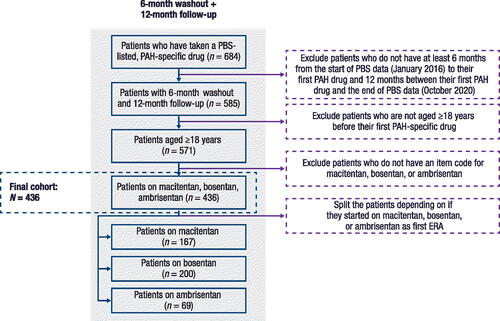
Table 1. Baseline demographic and clinical characteristics.
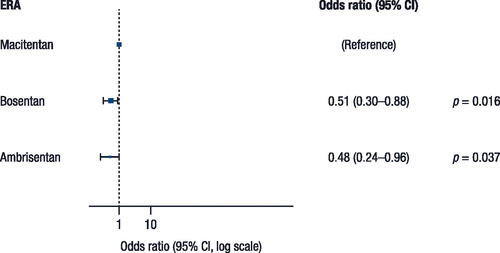
Figure 3. Sensitivity analyses for (A) 60% and (B) 70% adherence for all ERA treatments.
CI, confidence interval; ERA, endothelin receptor antagonist; PS, propensity score.
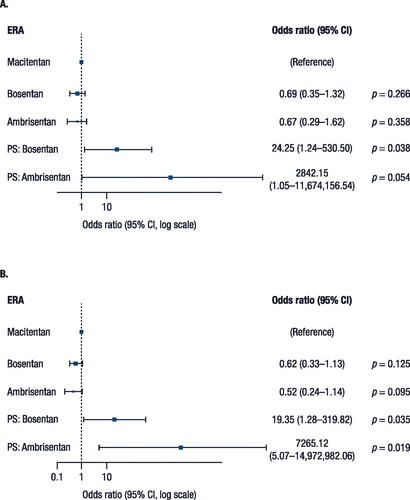
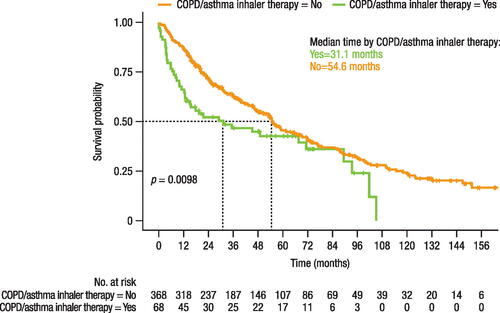
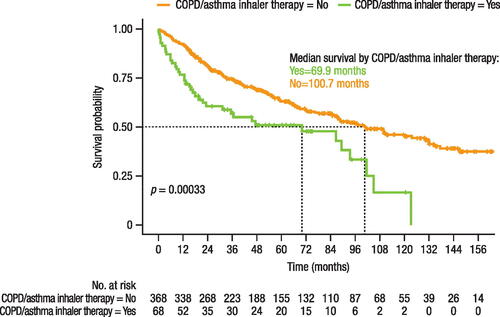
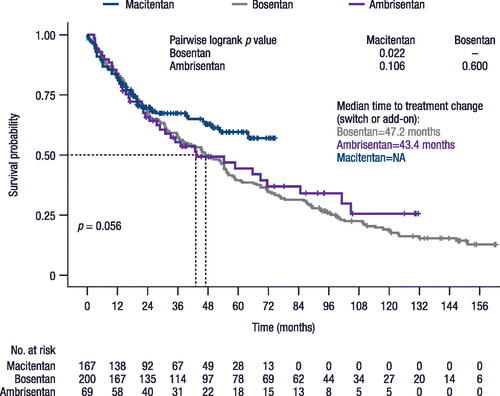
Figure 4. Time to treatment change (switch or add-on).
Note, time before switching may be impacted by the varying approval dates of the treatments (i.e. 2004 for bosentan, 2009 for ambrisentan, and 2014 for macitentan). NA, not achieved.
Supplemental Material
Download MS Word (295.8 KB)Data availability statement
The de-identified participant data will not be shared.