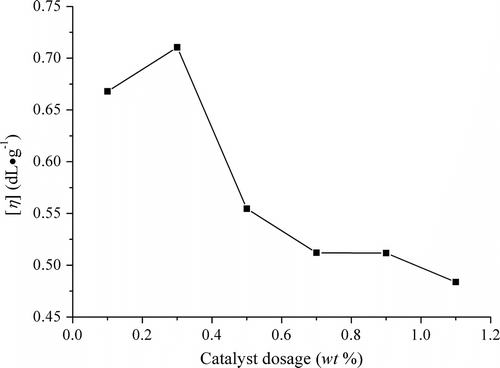
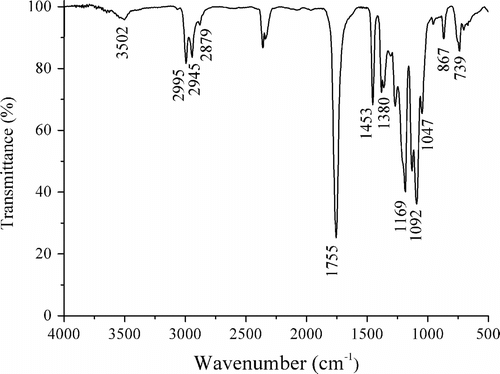
Figures & data
Table 1. The influences of different catalysts on the reaction.Footnote a
Figure 2 Influences of melt polymerization temperature on the viscosity of PLAB (conditions: 5 h and catalyst SnO dosage 0.3 wt%).
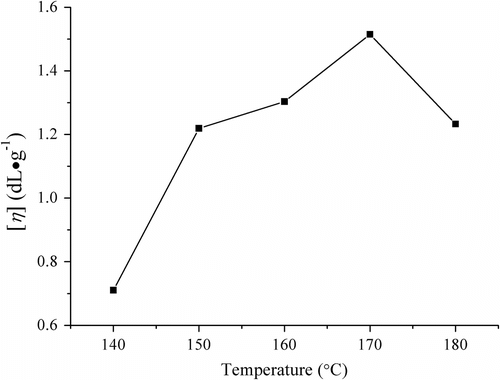
Figure 3 Influences of melt polymerization time on viscosity of PLAB (conditions: 170 °C and catalyst SnO dosage 0.3 wt%).
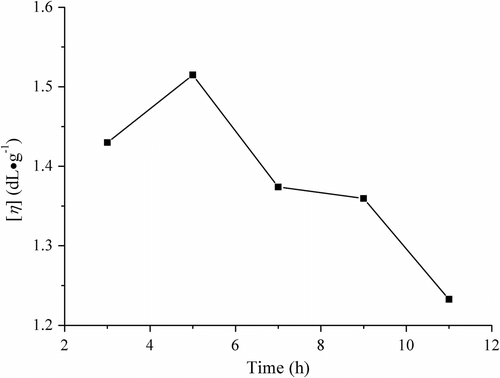
Table 2. The influences of molar feed ratios on yield, [η], and M n of the copolymers.Footnote a
Table 3. The influences of molar feed ratio on the thermal properties of PLAB.Footnote a
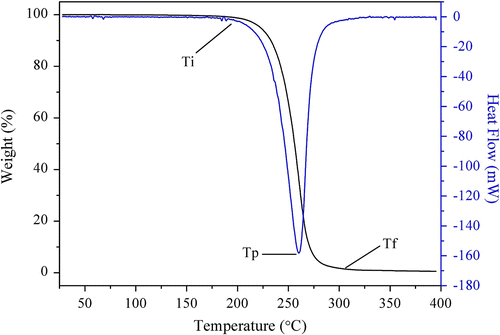
Figure 7 Differential thermogravimetry (DTG) curve of PLAB synthesized as the molar feed ratio 64/1 (LA/borneol).