Figures & data
Table 1. Chemical composition of hexane extract from Juniperus phoenicea leaves analyzed by GC-MS.
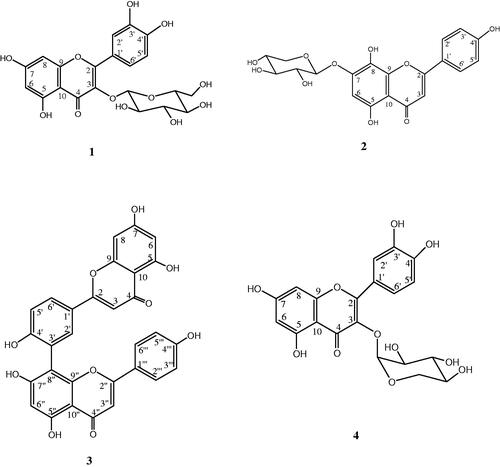
Figure 1. Chromatographic profiles at 330 nm of the fractions (Fr2, Fr3, and Fr4) of methanol extract. The numeration is that of .
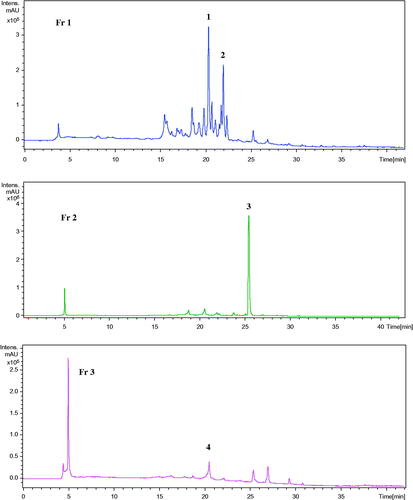
Table 2. Identification of the principal compounds in fractions Fr 1-3 by HPLC-MS-UV.
Figure 2. Nomenclature adopted for the retrocyclization fragments of biflavone (illustrated on amentoflavone).
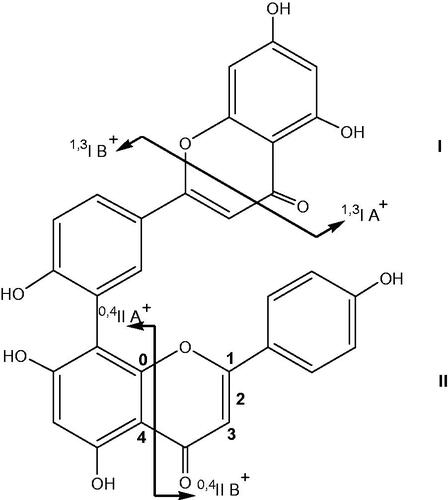
Table 3. Comparison of the 1H NMR spectroscopic data of compound 3 with those of amentoflavone and robustaflavone.
Table 4. DPPH-radical scavenging and α-amylase inhibition activities of hexane and methanolic extracts, fractions Fr1–3 and amentoflavone.