Figures & data
Figure 1. HPLC-TOF/MS chromatograms of the extracts from C. tinctoria: (a) ethyl acetate extract, (b) n-BuOH extract, (c) methanol-chloroform extract, (d) water extract.
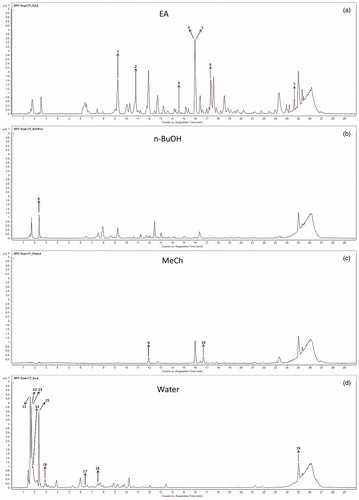
Figure 2. The main compounds from the extracts determined by HPLC/TOF-MS (a) ethyl acetate extract, (b) n-BuOH extract, (c) methanol-chloroform extract, (d) water extract.
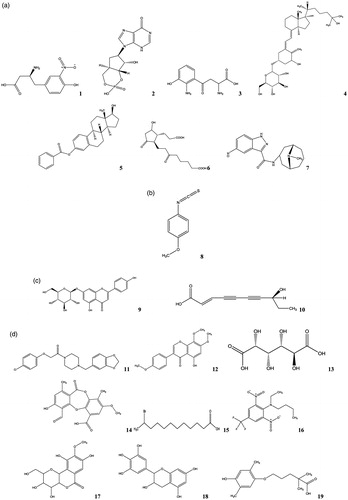
Table 1. Quantification of phenolic compounds of the extracts.
Figure 3. Antiproliferative effects of the ethyl acetate and butanol extracts (a) and active fractions (b) from C. tinctoria.
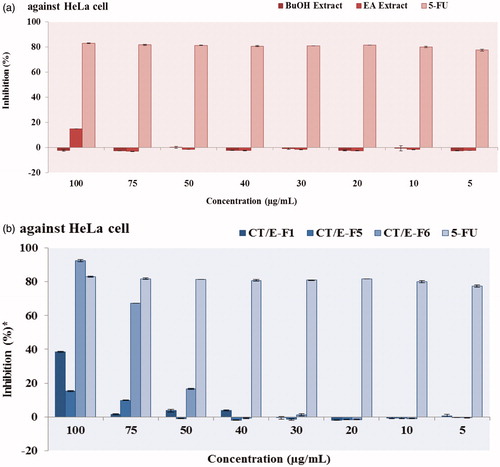
Table 2. The antiproliferative effects of C. tinctoria EA extract and obtained fractions at the tested concentrations.
Table 4. DPPH radical scavenging and antiproliferative effects of major phenolic compounds of EA extract.
Figure 4. DPPH radical scavenging of the EA extract, fractions, and BHT at the tested concentrations.
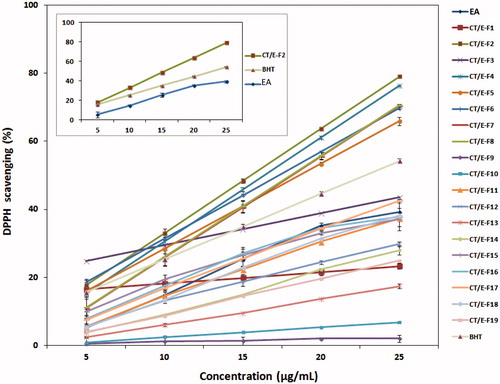
Table 3. Antioxidant activities of the fractions and ethyl acetate extract from C. tinctoriaTable Footnote*.
