Figures & data
Figure 1. The chromatograph chart at 354 nm (A) and the (−)-ESI total ion current (B) of Typha angustifolia.
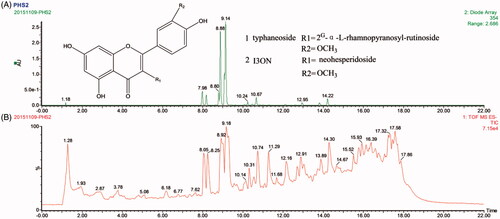
Table 1. The identification of flavone compounds in Typha angustifolia.
Figure 2. The DPPH scavenging activity (A) and Fe3+ absorbance (B) of the extract of Typha angustifolia.
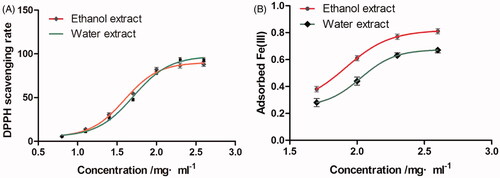
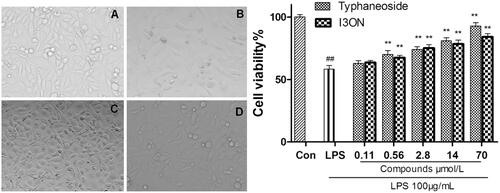
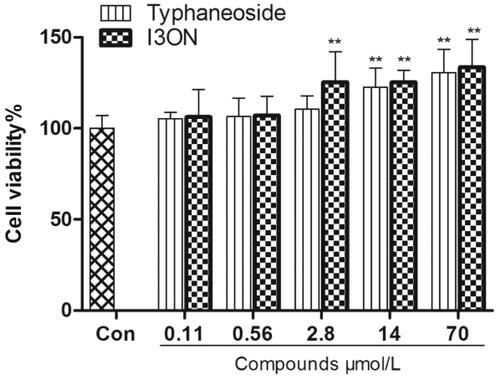
Figure 4. Effect of typhaneoside and I3ON on HUVECs stimulated with LPS. (A) normal control. (B) HUVECs was treated with of LPS (100 μg/mL) stimulation for 24 h. (C) HUVECs induced by LPS was treated with typhaneoside (70 μmol/L). (D) HUVECs induced by LPS was treated with I3ON (70 μmol/L), magnification ×200. Data are expressed as the means ± SD. (standard deviation, n = 5). #p < 0.05, ##p < 0.01 vs. sham control; *p < 0.05, **p < 0.01 vs. LPS group. Con: sham control group, LPS: LPS group.