Figures & data
Figure 1. Patient flowchart. Flowchart displaying the total number of patients screened for eligibility, the number of eligible patients, and the patients included in the study. One patient withdrew consent prior to intervention and was not included in data analyses. TA-TAVI: transapical transcatheter aortic valve implantation; SAVR: surgical aortic valve replacement.
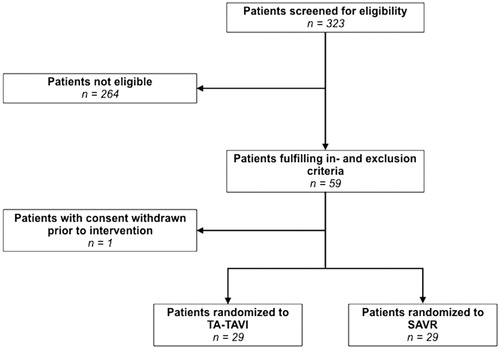
Table 1. Baseline characteristics of patients undergoing transapical transcatheter aortic valve implantation or surgical aortic valve replacement.
Figure 2. Quality-of life (SF-36). Graphical description of quality-of life (SF-36) divided in physical composite summary (A) and mental composite summary (B) during 5 years of follow-up in patients undergoing TA-TAVI (n = 29) or SAVR (n = 29). Horizontal lines indicate standard deviations. TA-TAVI: transapical transcatheter aortic valve implantation; SAVR: surgical aortic valve replacement.
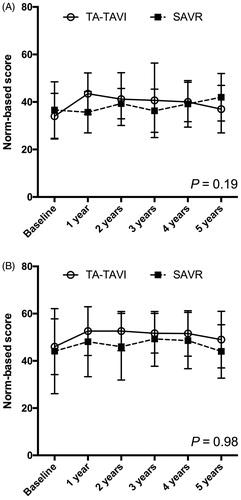
Table 2. Quality-of life (SF-36) in patients undergoing transapical transcatheter aortic valve implantation or surgical aortic valve replacement during 5 years of follow-up.
Table 3. Quality-of life (SF-36) in patients undergoing transapical transcatheter aortic valve implantation or surgical aortic valve replacement after 5 years of follow-up.
