Figures & data
Table 1. Baseline demographics and patient characteristics (safety and effectiveness analysis set).
Figure 1. Change in effectiveness measures over time. (a) DAS28-CRP. (b) DAS28-ESR. (c) CDAI. (d) SDAI. (e) HAQ-DI. (f) EQ-5D score. Data <1.0% are represented in the bars but are not labeled. CDAI: Clinical Disease Activity Index; DAS28-CRP: Disease Activity Score in 28 joints using C-reactive protein; DAS28-ESR: Disease Activity Score in 28 joints using erythrocyte sedimentation rate; EQ-5D: EuroQol-5 Dimensions; HAQ-DI: Health Assessment Questionnaire-Disability Index; SD: standard deviation; SDAI: Simplified Disease Activity Index.
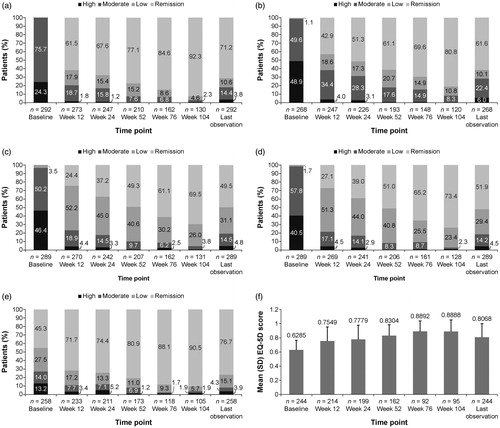
Figure 4. Change in effectiveness scores by MTX dose over time. (a) DAS28-CRP. (b) HAQ-DI. (c) mTSS. DAS28-CRP: Disease Activity Score in 28 joints using C-reactive protein; HAQ-DI: Health Assessment Questionnaire-Disability Index; mTSS: modified total Sharp score; MTX: methotrexate; SD: standard deviation.
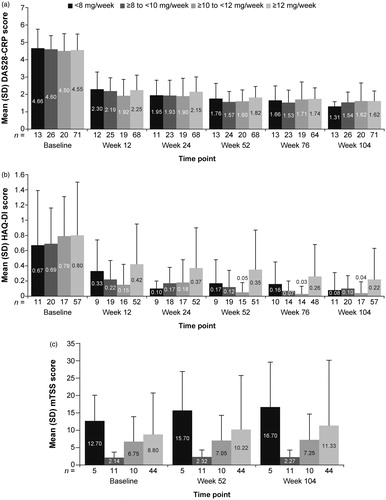
Table 2. ADRs in patients with or without methotrexate dose tapering.