Figures & data
Figure 1. Patient disposition. *Number of patients with a finalized demographic characteristics form. Note that some excluded patients had more than one reason for exclusion. eCRF, electronic case report form; ADR, adverse drug reaction.
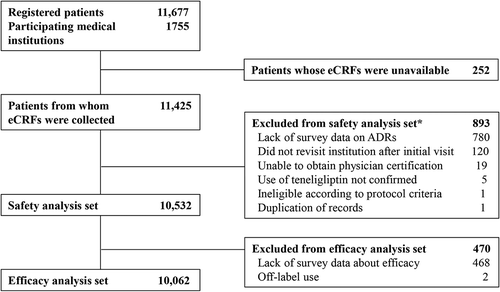
Table 1. Baseline characteristics (safety analysis set).
Table 2. Concomitant use of antidiabetic drugs during the survey period (safety analysis set).
Table 3. Incidence of ADRs and serious ADRs (safety analysis set).
Figure 2. Change in HbA1c from baseline to 2 years: (a) all patients (b) according to baseline HbA1c, in the efficacy analysis set. Data are expressed as the mean ± SD. The number of patients [N] at each point (a) and the number of patients and actual value (at baseline and after 2 years of treatment) in each group (b) are shown in parentheses. *p < 0.001 vs baseline, by paired-t test.
![Figure 2. Change in HbA1c from baseline to 2 years: (a) all patients (b) according to baseline HbA1c, in the efficacy analysis set. Data are expressed as the mean ± SD. The number of patients [N] at each point (a) and the number of patients and actual value (at baseline and after 2 years of treatment) in each group (b) are shown in parentheses. *p < 0.001 vs baseline, by paired-t test.](/cms/asset/6f3321d7-e9b1-4bb5-b2e5-792a733796d8/ieop_a_1420165_f0002_oc.jpg)

![Figure 3. LS-mean change in HbA1c from baseline to 2 years according to age group in the efficacy analysis set. Data are expressed as LS-Mean ± SE. The number of patients [N] at each point is shown in parentheses.](/cms/asset/742dd1b2-bfaf-4fb0-babb-a9d15cae53e6/ieop_a_1420165_f0003_oc.jpg)