Figures & data
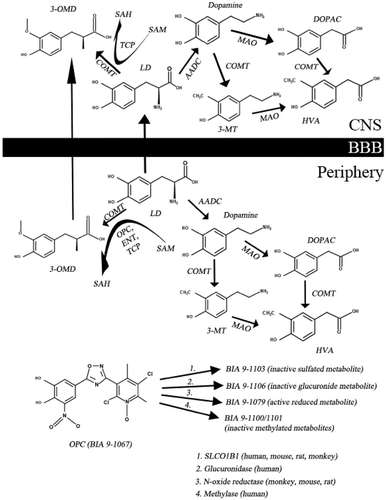
Table 1. Clinical trials with OPC (AUC – area under the curve; BIPARK – efficacy and safety of BIA 9–1067 in idiopathic Parkinson’s disease patients; BZ – benserazide; CD – carbidopa; Cmin – minimum blood plasma concentration; COMT – catechol-O-methyltransferase; ENT – entacapone; LD – levodopa; OPC – opicapone; PD – Parkinson’s disease; PLC – placebo). Adapted from [Citation14] with permission of Taylor & Francis. Data also derived from [Citation7] and [Citation10].