Figures & data
Table 1. Key TFQ clinical efficacy studies
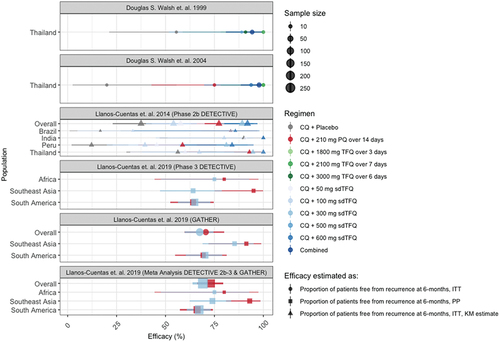
Figure 2. Efficacy of TFQ (x-axis) across radical cure trials (panels) and populations (y-axis). Shapes represent the method used to report efficacy of TFQ in preventing recurrences at 6-month follow. Colors represent the regimen used. Shape sizes represent the corresponding sample size. By region, study sites of the Phase 3 DETECTIVE trial were Ethiopia (Africa), Cambodia, Philippines, and Thailand (Asia), and Brazil and Peru (South America). By region, study sites of the Phase 3 GATHER trial were Thailand and Vietnam (Southeast Asia), and Brazil, Colombia and Peru (South America). Except for Phase 3 GATHER, where reported estimates were from Kaplan-Meier (KM) analysis, efficacy was estimated as proportion of patients free from recurrence at 6 months by Intention-To-Treat (ITT) analysis, or by Per-Protocol (PP) if ITT was not reported for the comparisons.
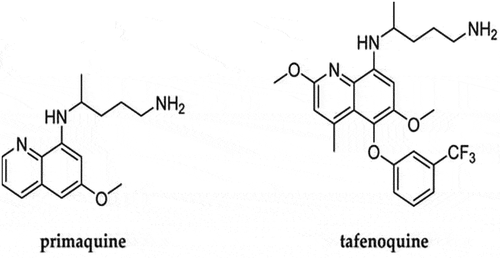
Drug Summary Box